G0535
Glycopeptidase A from almonds
buffered aqueous glycerol solution, ≥0.05 unit/mL
Synonym(s):
N-Glycosidase A, N-linked-glycopeptide-(N-acetyl-β-D-glucosaminyl)-L-asparagine amidohydrolase, PNGase A
About This Item
Recommended Products
conjugate
(N-linked)
Quality Level
form
buffered aqueous glycerol solution
concentration
≥0.05 unit/mL
storage temp.
−20°C
Related Categories
General description
Application
Biochem/physiol Actions
Unit Definition
Physical form
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
No data available
Flash Point(C)
No data available
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
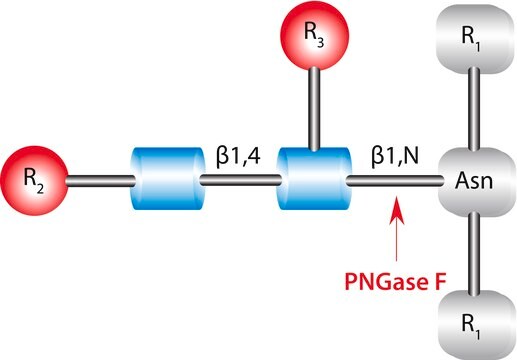
N-Linked Glycan Strategies. Use of the endoglycosidic enzyme PNGase F (N-Glycosidase F) is the most effective method of removing virtually all N-linked oligosaccharides from glycoproteins.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service