475913
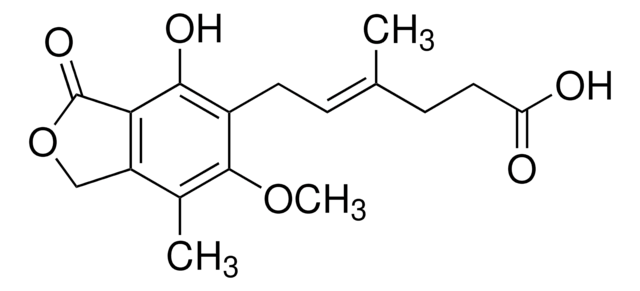
Mycophenolic Acid
An immunosuppressive agent that inhibits de novo purine nucleotide synthesis via inhibition of inosine monophosphate dehydrogenase and prevents the formation of XMP and GMP.
Synonym(s):
Mycophenolic Acid, 6-(4-Hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic Acid, MPA
About This Item
Recommended Products
Quality Level
Assay
≥95% (HPLC)
form
solid
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
color
white to off-white
solubility
0.1 M NaOH: 10 mg/mL
ethanol: soluble
shipped in
ambient
storage temp.
10-30°C
InChI
1S/C17H20O6/c1-9(5-7-13(18)19)4-6-11-15(20)14-12(8-23-17(14)21)10(2)16(11)22-3/h4,20H,5-8H2,1-3H3,(H,18,19)/b9-4+
InChI key
HPNSFSBZBAHARI-RUDMXATFSA-N
General description
Biochem/physiol Actions
Inosine monophosphate dehydrogenase
Warning
Reconstitution
Other Notes
Senda, M., et al. 1995. Transplantation60, 1143.
Warner-Bartnicki, A.L., et al. 1993. Exp. Cell Res.204, 241.
Falkner, F.G., and Moss, B. 1988. J. Virol.62, 1849.
Cohen, M.B., et al. 1981. J. Biol. Chem.256, 8713.
Mulligan, R.C., and Berg, P. 1981. Proc. Natl. Acad. Sci. USA78, 2072.
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Muta. 2 - Repr. 1B - STOT RE 1 Oral
Target Organs
Immune system
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service