D139459
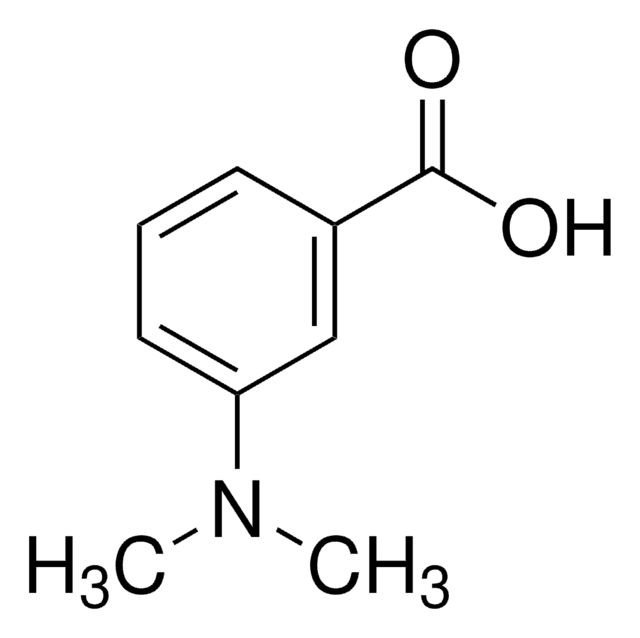
4-(Dimethylamino)benzoic acid
98%, for peptide synthesis
Synonym(s):
N,N-Dimethyl-4-aminobenzoic acid, N,N-Dimethyl-p-aminobenzoic acid, p-(Dimethylamino)benzoic acid
About This Item
Recommended Products
Product Name
4-(Dimethylamino)benzoic acid, 98%
Quality Level
Assay
98%
form
powder and chunks
reaction suitability
reaction type: solution phase peptide synthesis
mp
241-243 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
CN(C)c1ccc(cc1)C(O)=O
InChI
1S/C9H11NO2/c1-10(2)8-5-3-7(4-6-8)9(11)12/h3-6H,1-2H3,(H,11,12)
InChI key
YDIYEOMDOWUDTJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D139459-100G | 4061838353726 |
| D139459-25G | 4061833559758 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service