All Photos(1)
About This Item
Linear Formula:
BrC6H3(F)CN
CAS Number:
Molecular Weight:
200.01
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
76-80 °C (lit.)
SMILES string
Fc1ccc(Br)cc1C#N
InChI
1S/C7H3BrFN/c8-6-1-2-7(9)5(3-6)4-10/h1-3H
InChI key
GYCNHFWRPJXTSB-UHFFFAOYSA-N
Application
5-Bromo-2-fluorobenzonitrile may be used for the preparation of the following compounds:
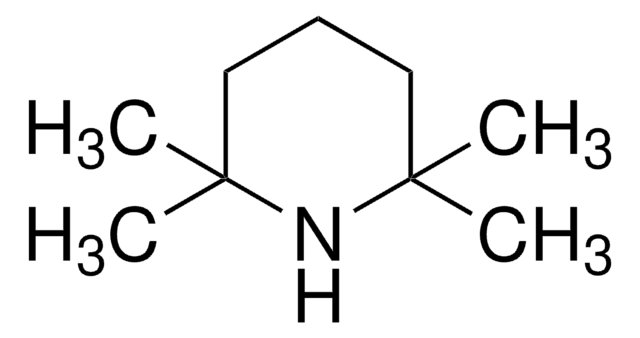
- (E)-5-(2-cyclopropylvinyl)-2-fluorobenzonitrile and methyl-3-amino-5-bromobenzo[b]thiophene-2-carboxylate
- (S)-[2-[5-(3-cyano-4-fluoro-phenyl)-pyridin-3-yloxy]-1-(1H-indol-3-ylmethyl)-ethyl]-carbamic acid tert-butyl ester
- 4-fluoro-3-cyano-3′-tributyltinbenzhydrol
- 4-fluoro-3-cyano-3′-iodobenzhydrol
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Microwave-assisted synthesis of 3-aminobenzo [b] thiophene scaffolds for the preparation of kinase inhibitors.
Bagley MC, et al.
Organic & Biomolecular Chemistry (2015)
Zizhong Li et al.
Bioconjugate chemistry, 14(2), 287-294 (2003-03-20)
The synthesis of a benzophenone-based labeling compound designed for comparative imaging studies with both in vivo positron emission tomograph (PET) and single-photon computed tomography (SPECT) and ex vivo autoradiography is described. The new compound can be labeled with either F-18
Keith W Woods et al.
Bioorganic & medicinal chemistry, 14(20), 6832-6846 (2006-07-18)
A series of heteroaryl-pyridine containing inhibitors of Akt are reported. The synthesis and structure-activity relationships are discussed, leading to the discovery of a indazole-pyridine analogue (K(i)=0.16 nM). These compounds bind in the ATP binding site, are potent, ATP competitive, and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)