All Photos(1)
About This Item
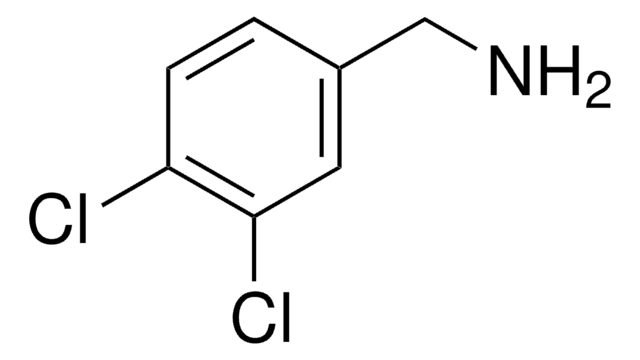
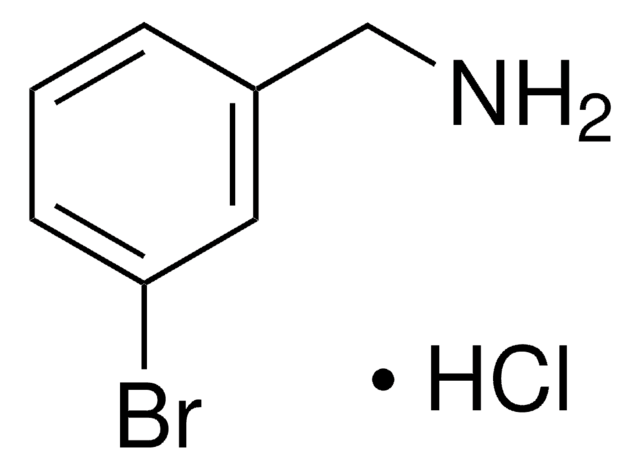
Linear Formula:
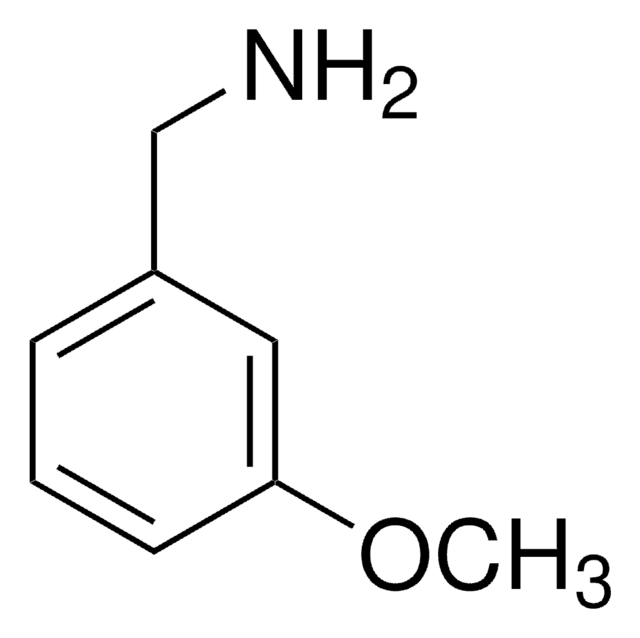
ClC6H4CH2NH2
CAS Number:
Molecular Weight:
141.60
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
liquid
refractive index
n20/D 1.559 (lit.)
bp
110-112 °C/17 mmHg (lit.)
density
1.159 g/mL at 25 °C (lit.)
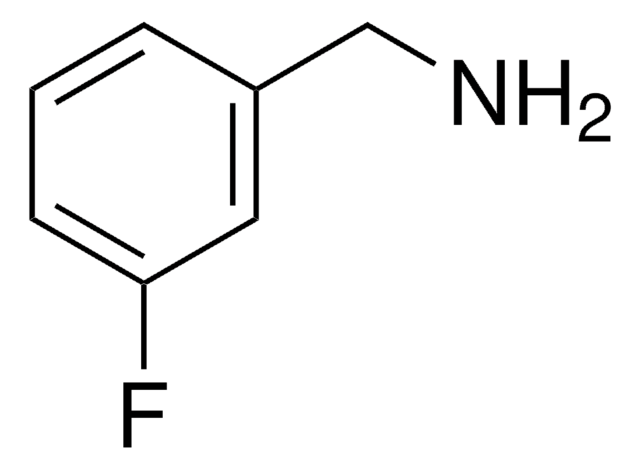
SMILES string
NCc1cccc(Cl)c1
InChI
1S/C7H8ClN/c8-7-3-1-2-6(4-7)5-9/h1-4H,5,9H2
InChI key
BJFPYGGTDAYECS-UHFFFAOYSA-N
General description
3-Chlorobenzylamine undergoes reductive amination during dihydroquinolone synthesis. It is used in the synthesis of N- (3-chlorobenzyl) toluene-p-sulphonamide.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
208.4 °F - closed cup
Flash Point(C)
98 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Stefan Jaroch et al.
Bioorganic & medicinal chemistry letters, 13(12), 1981-1984 (2003-06-05)
Dihydroquinolines with aminoalkyl side chains have been synthesized and have been shown to be potent n-NOS inhibitors. A marked selectivity versus e-NOS of up to approximately 300-fold was observed, whereas i-NOS was moderately inhibited.
Rates of reaction of benzylamines with 1-chloro-2, 4-dinitrobenzene and with toluene-p-sulphonyl chloride.
Journal of the Chemical Society B: Physical Organic, 466-468 (1966)
E Farcaş et al.
Analytica chimica acta, 1034, 214-222 (2018-09-09)
Only few reports describe the use of capillary electrophoresis in the context of Fragment Based Drug Discovery (FBDD). In this paper, we will present a generic, fully automated, microscale electrophoretic mobility shift displacement assay that can be used in FBDD
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service