G1774
Glucagon
≥95% (HPLC), powder, synthetic
About This Item
Produits recommandés
Source biologique
synthetic
Niveau de qualité
Stérilité
non-sterile
Pureté
≥95% (HPLC)
Forme
powder
Technique(s)
cell based assay: suitable
Solubilité
1% acetic acid: 1.00-1.04 mg/mL, clear, colorless
water: 1.00-1.04 mg/mL, clear, colorless
Adéquation
suitable for molecular biology
Numéro d'accès UniProt
Conditions d'expédition
ambient
Température de stockage
−20°C
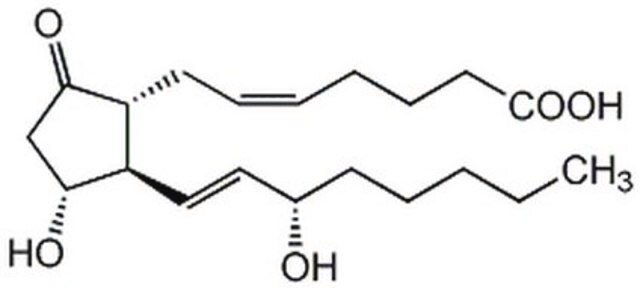
Chaîne SMILES
Cl[H].[H]N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)NCC(=O)N[C@@H](C(C)O)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](C(C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc3ccc(O)cc3)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc4ccc(O)cc4)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc5ccccc5)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](Cc6c[nH]c7ccccc67)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)O)C(O)=O
InChI
1S/C153H225N43O49S.ClH/c1-72(2)52-97(133(226)176-96(47-51-246-11)132(225)184-104(60-115(159)209)143(236)196-123(78(10)203)151(244)245)179-137(230)103(58-83-64-167-89-29-19-18-28-87(83)89)183-131(224)95(43-46-114(158)208)177-148(241)120(74(5)6)194-141(234)101(54-79-24-14-12-15-25-79)182-138(231)105(61-117(211)212)185-130(223)94(42-45-113(157)207)171-124(217)75(7)170-127(220)91(31-22-49-165-152(160)161)172-128(221)92(32-23-50-166-153(162)163)174-146(239)110(69-199)191-140(233)107(63-119(215)216)186-134(227)98(53-73(3)4)178-135(228)99(56-81-33-37-85(204)38-34-81)180-129(222)90(30-20-21-48-154)173-145(238)109(68-198)190-136(229)100(57-82-35-39-86(205)40-36-82)181-139(232)106(62-118(213)214)187-147(240)111(70-200)192-150(243)122(77(9)202)195-142(235)102(55-80-26-16-13-17-27-80)188-149(242)121(76(8)201)193-116(210)66-168-126(219)93(41-44-112(156)206)175-144(237)108(67-197)189-125(218)88(155)59-84-65-164-71-169-84;/h12-19,24-29,33-40,64-65,71-78,88,90-111,120-123,167,197-205H,20-23,30-32,41-63,66-70,154-155H2,1-11H3,(H2,156,206)(H2,157,207)(H2,158,208)(H2,159,209)(H,164,169)(H,168,219)(H,170,220)(H,171,217)(H,172,221)(H,173,238)(H,174,239)(H,175,237)(H,176,226)(H,177,241)(H,178,228)(H,179,230)(H,180,222)(H,181,232)(H,182,231)(H,183,224)(H,184,225)(H,185,223)(H,186,227)(H,187,240)(H,188,242)(H,189,218)(H,190,229)(H,191,233)(H,192,243)(H,193,210)(H,194,234)(H,195,235)(H,196,236)(H,211,212)(H,213,214)(H,215,216)(H,244,245)(H4,160,161,165)(H4,162,163,166);1H/t75-,76+,77+,78+,88-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,120-,121-,122-,123-;/m0./s1
Clé InChI
RKGLLHCSSVJTAN-YYICOITRSA-N
Informations sur le gène
human ... GCG(2641)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
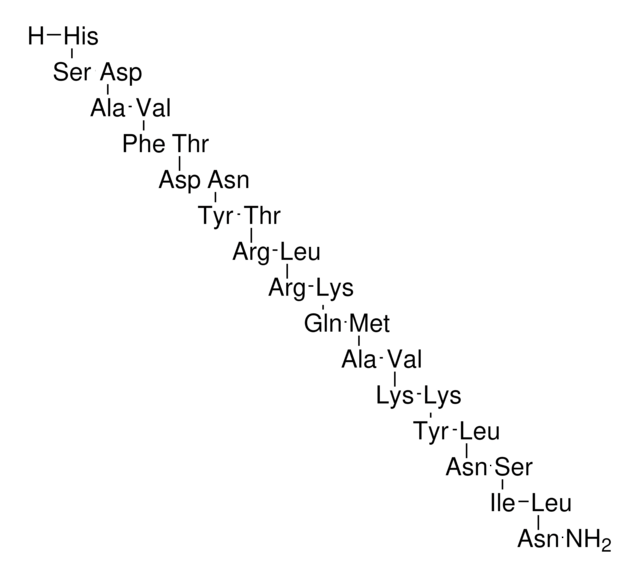
Amino Acid Sequence
Description générale
Application
- used as a component of hormone stock solution for preserving full biological activity of heart tissues obtained from male Sprague-Dawley rats
- used as an infusion in phases II and III fasting king penguins to study the various lipolytic, metabolic, and hormonal responses
- used for the stimulation of PGC-1α expression in hepatocytes
- used to induce the expression of methionine adenosyltransferase α1 (MAT1A), which is involved in the regulation of hepatic levels of S-adenosylmethionine and the adaptive response to fasting
- used to study its effect on stimulation of gluconeogenesis through hepatic lipolysis, mediated by inositol trisphosphate receptor 1 (INSP3R1)
- intraperitoneally injected in mice to assess glucagon-induced Sam68 subcellular localization in vivo
Actions biochimiques/physiologiques
Composants
Autres remarques
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique


![[Arg8]-Vasopressin solution Grade VI (synthetic), ~100 IU/mL in 0.9% NaCl](/deepweb/assets/sigmaaldrich/product/structures/326/242/dede8c26-cf73-4a28-a5d9-1d57c673cf0e/640/dede8c26-cf73-4a28-a5d9-1d57c673cf0e.png)