D5794
Diacylglycerol Kinase Inhibitor II
solid
Synonyme(s) :
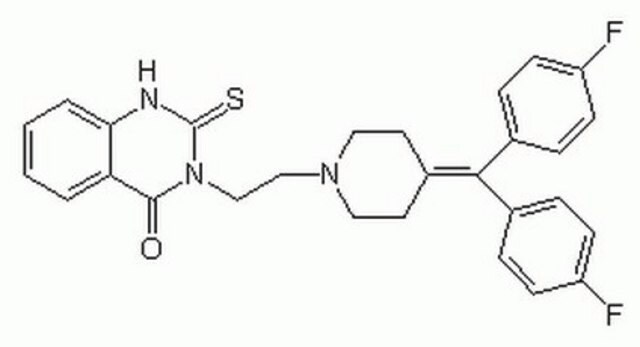
3-[2-[4-(bis(4-Fluorophenyl)methylene)-1-piperidinyl]ethyl]-2,3-dihydro-2-thioxo-4(1H)-quinazolinone, R59949
About This Item
Produits recommandés
Source biologique
synthetic (organic)
Essai
≥97% (HPLC)
Forme
solid
Couleur
pale yellow
Pf
228-230 °C
Solubilité
0.1 M HCl: slightly soluble
0.1 M NaOH: slightly soluble
DMSO: soluble
H2O: insoluble
ethanol: soluble
ethyl acetate: soluble
Température de stockage
−20°C
Chaîne SMILES
Fc1ccc(cc1)\C(=C2\CCN(CCN3C(=S)Nc4ccccc4C3=O)CC2)c5ccc(F)cc5
InChI
1S/C28H25F2N3OS/c29-22-9-5-19(6-10-22)26(20-7-11-23(30)12-8-20)21-13-15-32(16-14-21)17-18-33-27(34)24-3-1-2-4-25(24)31-28(33)35/h1-12H,13-18H2,(H,31,35)
Clé InChI
ZCNBZFRECRPCKU-UHFFFAOYSA-N
Catégories apparentées
Application
Actions biochimiques/physiologiques
Caractéristiques et avantages
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique