17793
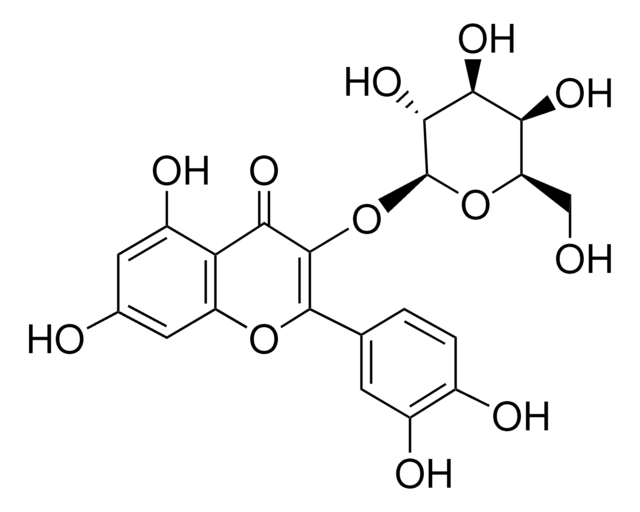
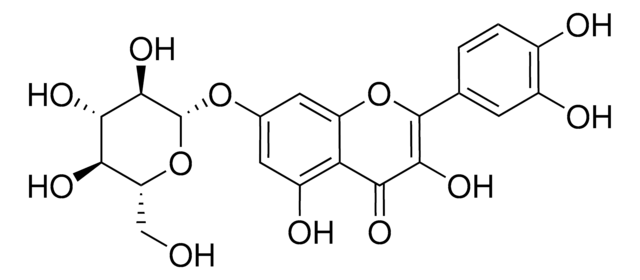
Quercetin 3-β-D-glucoside
≥90% (HPLC)
Synonyme(s) :
3,3′,4′,5,7-Pentahydroxyflavone 3-β-glucoside, Isoquercitrin
About This Item
Produits recommandés
Niveau de qualité
Pureté
≥90% (HPLC)
Application(s)
metabolomics
vitamins, nutraceuticals, and natural products
Température de stockage
−20°C
Chaîne SMILES
OC[C@H]1O[C@@H](OC2=C(Oc3cc(O)cc(O)c3C2=O)c4ccc(O)c(O)c4)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C21H20O12/c22-6-13-15(27)17(29)18(30)21(32-13)33-20-16(28)14-11(26)4-8(23)5-12(14)31-19(20)7-1-2-9(24)10(25)3-7/h1-5,13,15,17-18,21-27,29-30H,6H2/t13-,15-,17+,18-,21+/m1/s1
Clé InChI
OVSQVDMCBVZWGM-QSOFNFLRSA-N
Informations sur le gène
mouse ... Hexa(15211)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- as a dietary flavonoid supplement to check its binding capacity with human small ubiquitin-related modifier 1 (SUMO1) protein using surface plasmon resonance (SPR)
- as an inhibitor for Escherichia coli adenosine triphosphate (ATP) synthase
- as an anti-aggregation agent to test its activity against β-amyloid, green fluorescent protein (GFP), and chymotrypsinogen proteins
Actions biochimiques/physiologiques
Conditionnement
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Protocoles
HPLC Analysis of Polyphenols in Nero d'Avola Red Wine on Discovery® HS C18 (UV 280 nm)
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique