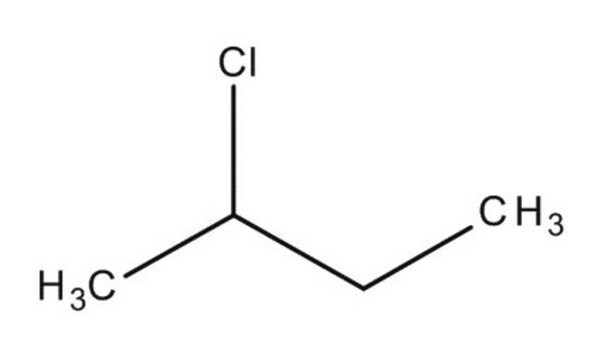
414255
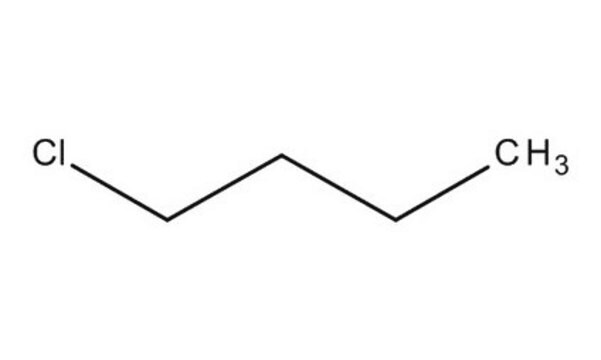
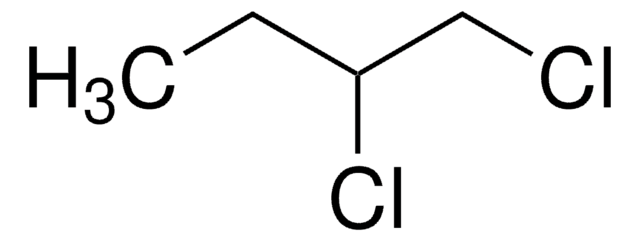
1-Chlorobutane
anhydrous, 99.5%
Synonym(s):
Butyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
CH3(CH2)3Cl
CAS Number:
Molecular Weight:
92.57
Beilstein:
1730909
EC Number:
MDL number:
UNSPSC Code:
12352101
PubChem Substance ID:
NACRES:
NA.21
Recommended Products
grade
anhydrous
Quality Level
vapor density
3.2 (vs air)
vapor pressure
80.1 mmHg ( 78.4 °C)
Assay
99.5%
form
liquid
autoignition temp.
860 °F
expl. lim.
10.1 %
impurities
<0.002% water
evapn. residue
<0.0003%
refractive index
n20/D 1.402 (lit.)
bp
77-78 °C (lit.)
mp
−123 °C (lit.)
density
0.886 g/mL at 25 °C (lit.)
SMILES string
CCCCCl
InChI
1S/C4H9Cl/c1-2-3-4-5/h2-4H2,1H3
InChI key
VFWCMGCRMGJXDK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
1-Chlorobutane is an isomer of chlorobutane. It undergoes stoichiometric and catalytic dehydrochlorination on reacting with nanocrystalline MgO (magnesium oxide) to generate isomers of butene. Viscosities of the binary mixtures of pentyl acetate and 1-chlorobutane and 1-chlorobutane and acetonitrile has been studied. This study helps in better understanding of cosolvency in polymer system.
Application
1-Chlorobutane may be used in the synthesis of 1-butyl-3-methylimidazolium hydrogen sulfate ([BMIM][HSO4]).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Asp. Tox. 1 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
10.4 °F - closed cup
Flash Point(C)
-12 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Extraction and oxidative desulfurization of diesel fuel catalyzed by a Br?nsted acidic ionic liquid at room temperature.
Gao H, et al.
Green Chemistry, 12(7), 1220-1224 (2010)
Viscosities and excess volumes of binary mixtures formed by the liquids acetonitrile, pentyl acetate, 1-chlorobutane, and carbon tetrachloride at 25?C.
Prolongo MG, et al.
The Journal of Physical Chemistry, 88(10), 2163-2167 (1984)
Simultaneous Prediction of Densities and Vapor-Liquid Equilibria of Mixtures Containing an Isomeric Chlorobutane and Methyl tert-Butyl Ether Using the VTPR Model.
Montano D, et al.
Industrial & Engineering Chemistry Research, 50(24), 14193-14202 (2011)
Changes in texture and catalytic activity of nanocrystalline MgO during its transformation to MgCl2 in the reaction with 1-chlorobutane.
Fenelonov VB, et al.
The Journal of Physical Chemistry B, 105(18), 3937-3941 (2001)
Antonio García Martínez et al.
The Journal of organic chemistry, 70(25), 10238-10246 (2005-12-06)
[structure: see text] The activation energy in the gas phase (deltaE(double dagger)) and the free energy of activation (deltaG(double dagger)) in water solution for the hydrolysis of the monohydrates of methyl chloride (MeCl), tert-butyl chloride (t-BuCl), and 1-adamantyl chloride (AdCl)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service