01-3540
6-Aminocaproic acid
SAJ special grade, ≥99.0%
Synonym(s):
ε-Aminocaproic acid, 6-Aminohexanoic acid, EACA
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
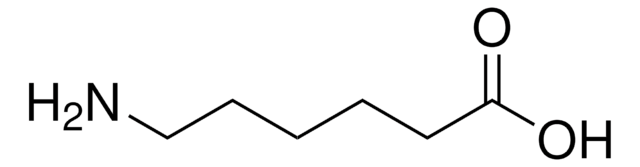
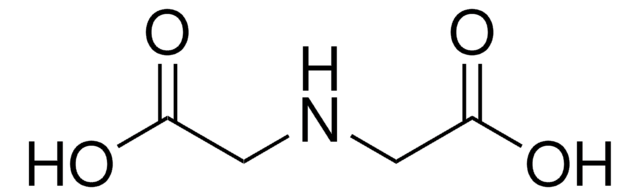
Linear Formula:
H2N(CH2)5CO2H
CAS Number:
Molecular Weight:
131.17
Beilstein:
906872
EC Number:
MDL number:
UNSPSC Code:
12352116
PubChem Substance ID:
Recommended Products
grade
SAJ special grade
Assay
≥99.0%
form
solid
availability
available only in Japan
mp
207 °C
207-209 °C (dec.) (lit.)
solubility
water: soluble
application(s)
peptide synthesis
SMILES string
NCCCCCC(O)=O
InChI
1S/C6H13NO2/c7-5-3-1-2-4-6(8)9/h1-5,7H2,(H,8,9)
InChI key
SLXKOJJOQWFEFD-UHFFFAOYSA-N
Gene Information
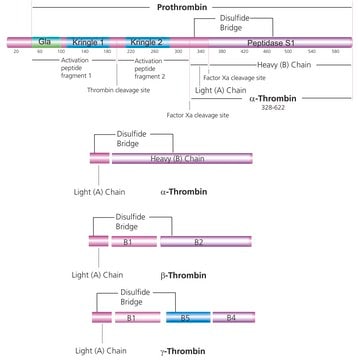
human ... PLAT(5327) , PLG(5340)
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
Lysine analog. Promotes rapid dissociation of plasmin, thereby inhibiting the activation of plasminogen and subsequent fibrinolysis. Reported to inhibit plasminogen binding to activated platelets. An early report indicated that it inhibits the activation of the first component of the complement system. Binds and inactivates Carboxypeptidase B.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Illa Ramakanth et al.
The journal of physical chemistry. B, 116(9), 2722-2729 (2012-02-09)
A two-component gel resulting from the amphiphilic cationic surfactant cetylpyridinium chloride (CPC) in the presence of a structure-forming bola-amphiphilic additive 6-aminocaproic acid (6-ACA) was realized and investigated. At a critical 6 wt % of 1:1 CPC:6-ACA, the yellow colored gel
Andrew B Jackson et al.
Nuclear medicine and biology, 39(3), 377-387 (2012-01-10)
The present study describes the design and development of a new heterodimeric RGD-bombesin (BBN) agonist peptide ligand for dual receptor targeting of the form (64)Cu-NO2A-RGD-Glu-6-Ahx-BBN(7-14)NH(2) in which Cu-64=a positron emitting radiometal; NO2A=1,4,7-triazacyclononane-1,4-diacetic acid; Glu=glutamic acid; 6-Ahx=6-aminohexanoic acid; RGD=the amino acid
Brian Hutton et al.
BMJ (Clinical research ed.), 345, e5798-e5798 (2012-09-13)
To estimate the relative risks of death, myocardial infarction, stroke, and renal failure or dysfunction between antifibrinolytics and no treatment following the suspension of aprotinin from the market in 2008 for safety reasons and its recent reintroduction in Europe and
Lajos Gera et al.
Peptides, 34(2), 433-446 (2012-02-22)
Peptide agonists and antagonists of both bradykinin (BK) B(1) and B(2) receptors (B(1)R, B(2)R) are known to tolerate to a certain level N-terminal sequence extensions. Using this strategy, we produced and characterized the full set of fluorescent ligands by extending
Ai Kia Yip et al.
Biophysical journal, 104(1), 19-29 (2013-01-22)
Cells sense the rigidity of their substrate; however, little is known about the physical variables that determine their response to this rigidity. Here, we report traction stress measurements carried out using fibroblasts on polyacrylamide gels with Young's moduli ranging from
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service