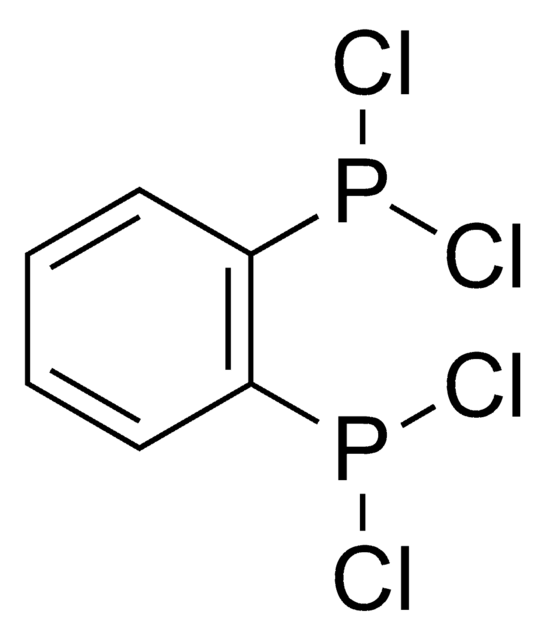
460273
1,2-Bis(diphenylphosphino)benzene
97%
Synonym(s):
1,2-Bis(diphenylphosphanyl)benzene, dppBz, dppbe, dppben, dppbenz, dppbz, o-Phenylenebis[diphenylphosphine], o-bis(diphenylphosphino)benzene
About This Item
Recommended Products
Assay
97%
form
solid
reaction suitability
reagent type: ligand
reaction type: Baeyer-Villiger Oxidation
reagent type: ligand
reaction type: Cycloadditions
reagent type: ligand
reaction type: Reductions
mp
183-188 °C (lit.)
functional group
phosphine
SMILES string
c1ccc(cc1)P(c2ccccc2)c3ccccc3P(c4ccccc4)c5ccccc5
InChI
1S/C30H24P2/c1-5-15-25(16-6-1)31(26-17-7-2-8-18-26)29-23-13-14-24-30(29)32(27-19-9-3-10-20-27)28-21-11-4-12-22-28/h1-24H
InChI key
NFRYVRNCDXULEX-UHFFFAOYSA-N
Application
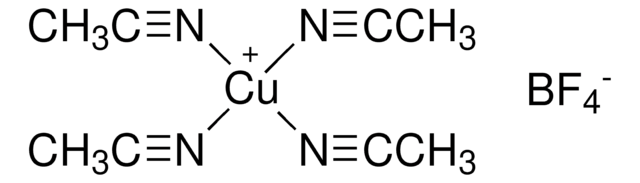
- As a chelating ligand in the synthesis of luminescent copper(I) halide complexes.
- In the synthesis of copper-1,2-bis(diphenylphosphino)benzene catalyst which is used for the β-boration of α, β-unsaturated amide.
- As an alternative to TMEDA for the selective cleavage of sp3 C-X bond in fluoroaromatic coupling reactions catalyzed by iron.
- As a ligand in the synthesis of alkenylboronates from acetylenic esters.
- To synthesize copper-diphosphine complexes which are used as homogeneous catalysts for N-formylation of a wide range of amines.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
The Baeyer-Villiger oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts the ketones to esters and the cyclic ketones to lactones.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![[1,2-Bis(diphenylphosphino)ethane]dichloronickel(II)](/deepweb/assets/sigmaaldrich/product/structures/707/956/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf/640/483e7a6e-5fb5-4e39-abd1-ecf33ccab3cf.png)