All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H7NO3
CAS Number:
Molecular Weight:
225.20
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
mp
222-223 °C (lit.)
functional group
ketone
nitro
SMILES string
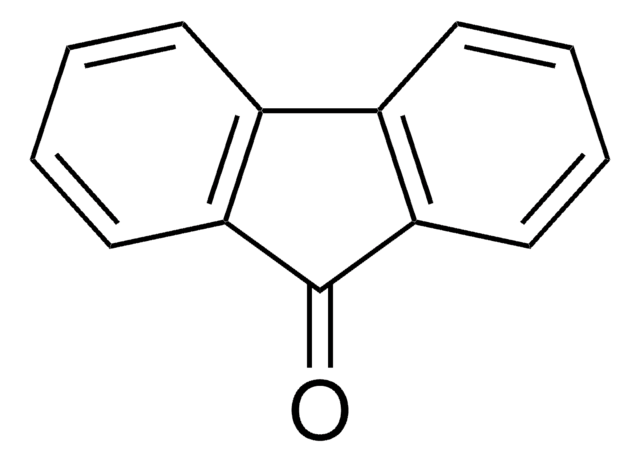
[O-][N+](=O)c1ccc2-c3ccccc3C(=O)c2c1
InChI
1S/C13H7NO3/c15-13-11-4-2-1-3-9(11)10-6-5-8(14(16)17)7-12(10)13/h1-7H
InChI key
AJEAHBZZHSLIQP-UHFFFAOYSA-N
Related Categories
General description
2-Nitro-9-fluorenone is a mutagenic photoproduct of u.v.a.-irradiated 2-aminofluorene. 2-Nitro-9-fluorenone was isolated from diesel-exhaust particles using a two-step fractionation scheme consisting of Sephadex LH20 chromatography and silica-gel thin-layer chromatography. Submicromolar concentrations of 2-nitro-9-fluorenone were quantitated by mercury meniscus modified silver solid amalgam electrode combined with direct current voltammetry or differential pulse voltammetry.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
S Itoh et al.
Biochemistry, 30(22), 5340-5346 (1991-06-04)
One-carbonyl quinonoid compounds, fluorenone (fluoren-9-one), anthrone, and their derivatives are introduced into spinach photosystem (PS) I reaction centers in place of the intrinsic secondary electron acceptor phylloquinone (= vitamin K1). Anthrone and 2-nitrofluorenone fully mediated the electron-transfer reaction between the
B Henkel et al.
Journal of peptide science : an official publication of the European Peptide Society, 7(3), 152-156 (2001-04-12)
This paper presents a new protecting group, the (2-nitrofluoren-9-yl)methoxycarbonyl group. Investigations on the properties of this new modification of the Fmoc-system, such as the solvent-dependent photochemical cleavage, and enhanced lability towards bases, are described, as well as UV-kinetic measurements of
Kari Kreander et al.
The Journal of pharmacy and pharmacology, 58(11), 1545-1552 (2006-11-30)
This study investigated the mutagenic, anti-mutagenic and cytotoxic effects of acetone extract of raspberry, Rubus idaeus L. (v. Ottawa) Rosaceae, and the isolated and characterized ellagitannin and anthocyanin fractions thereof, suitable for food applications. The studied raspberry extract and fractions
W E Bechtold et al.
Mutation research, 173(2), 105-109 (1986-02-01)
Organic extracts of diesel-exhaust particles show direct mutagenic activity in the Salmonella typhimurium bacterial mutagenicity assay. Nitro-aromatic compounds are believed to be responsible for part of the mutagenicity. A previously unidentified polyfunctional nitro-aromatic compound, 2-nitro-9-fluorenone (2N-Fone) was isolated from diesel-exhaust
G F Strniste et al.
Carcinogenesis, 7(3), 499-502 (1986-03-01)
Exposure of solutions of 2-aminofluorene (2-AF, dissolved in dimethylsulfoxide) to near ultraviolet light (u.v.a. wavelengths of 320-400 nm) results in the formation of a variety of photo-products, several of which are direct-acting mutagens in the Ames/Salmonella standard-plate assay. Previously published
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service