284270
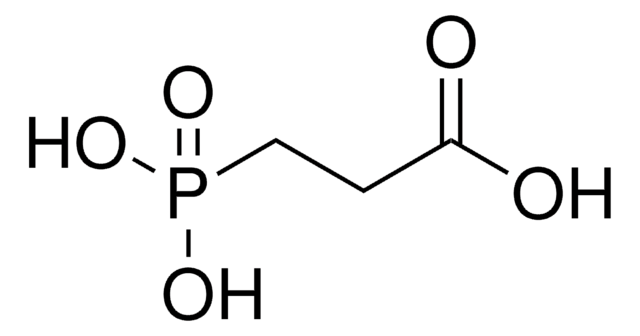
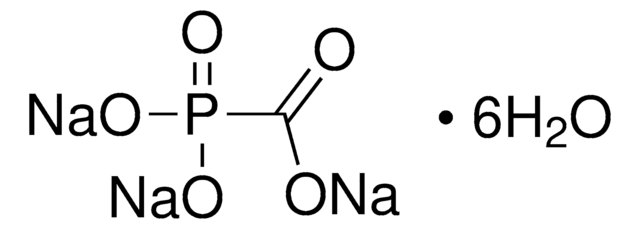
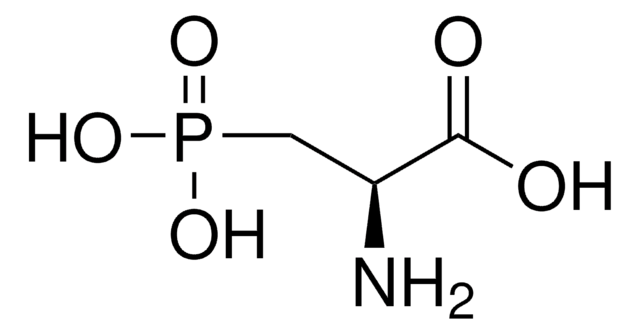
Phosphonoacetic acid
98%
Synonym(s):
(Carboxymethyl)phosphonic acid, 2-Phosphonoacetic acid, Fosfonoacetic acid, Phosphonacetic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
(HO)2P(O)CH2CO2H
CAS Number:
Molecular Weight:
140.03
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
powder or crystals
mp
143-146 °C (lit.)
solubility
water: soluble 100 mg/mL, clear to very slightly hazy, colorless
functional group
carboxylic acid
SMILES string
OC(=O)CP(O)(O)=O
InChI
1S/C2H5O5P/c3-2(4)1-8(5,6)7/h1H2,(H,3,4)(H2,5,6,7)
InChI key
XUYJLQHKOGNDPB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Phosphonoacetic acid inhibits specifically human cytomegalovirus DNA synthesis in virus-infected human fibroblasts. It also inhibits the synthesis of herpes simplex virus DNA in infected cells and the activity of the virus-specific DNA polymerase in vitro.
Application
Phosphonoacetic acid was used as an inhibitor of viral DNA replication, to investigate the infected cell protein 0 (bICP0) localization in infected low passage bovine cells. It was also used as a phosphorus source for microbial growth in phosphate-independent manner.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
R W Honess et al.
Journal of virology, 21(2), 584-600 (1977-02-01)
Phosphonoacetic acid (PAA) inhibited the synthesis of herpes simplex virus DNA in infected cells and the activity of the virus-specific DNA polymerase in vitro. In the presence of concentrations of PAA sufficient to prevent virus growth and virus DNA synthesis
Alireza Mahmoudian et al.
Comparative immunology, microbiology and infectious diseases, 35(2), 103-115 (2011-12-27)
The kinetics of expression of only a few genes of infectious laryngotracheitis virus (ILTV) have been determined, using northern blot analysis. We used quantitative reverse transcriptase PCR to examine the kinetics of expression of 74 ILTV genes in LMH cells.
E S Huang
Journal of virology, 16(6), 1560-1565 (1975-12-01)
Phosphonoacetic acid specifically inhibited human cytomegalovirus DNA synthesis in virus-infected human fibroblasts as detected by virus-specific nucleic acid hybridization. Inhibition was reversible; viral DNA synthesis resumed upon the removal of the drug. The compound partially inhibited DNA synthesis of host
Jacqueline P Smits et al.
The Journal of organic chemistry, 76(21), 8807-8813 (2011-09-16)
The α-trisphosphonic acid esters provide a unique spatial arrangement of three phosphonate groups and may represent an attractive motif for inhibitors of enzymes that utilize di- or triphosphate substrates. To advance studies of this unique functionality, a general route to
Magdalena Klimek-Ochab
Folia microbiologica, 59(5), 375-380 (2014-02-27)
A psychrophilic fungal strain of Geomyces pannorum P15 was screened for its ability to utilize a range of synthetic and natural organophosphonate compounds as the sole source of phosphorus, nitrogen, or carbon. Only phosphonoacetic acid served as a phosphorus source
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 284270-10G | 4061826253342 |
| 284270-50G | 4061833545911 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service