SML1959
GA3-AM
≥95% (HPLC)
Synonym(e):
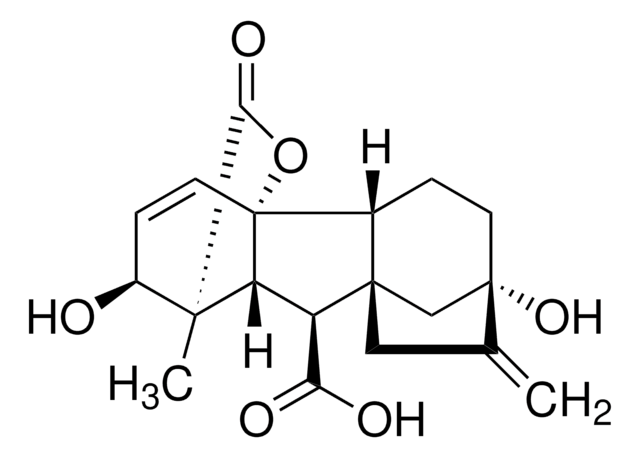
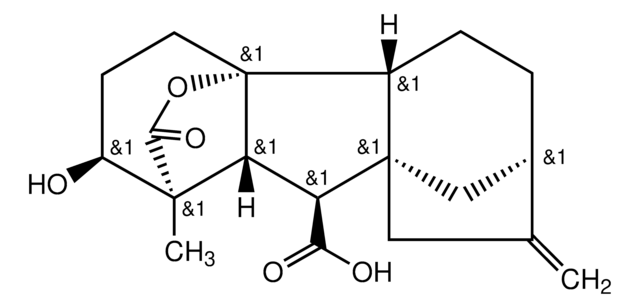
(1α,2β,4aα,4bβ,10β)-2,4a,7-Trihydroxy-1-methyl-8-methylenegibb-3-ene-1,10-dicarboxylic Acid 1,4a-Lactone Acetoxymethyl Ester, (1S,2S,4aR,4bR,7S,9aS,10S,10aR)-1,2,4b,5,6,7,8,9,10,10a-decahydro-2,7-dihydroxy-1-methyl-8-methylene-13-oxo-4a,1-(Epoxymethano)-7,9a-methanobenz[a]azulene-10-acetic acid (acetyloxy)methyl ester, Gibberellic Acid Acetoxymethyl Ester
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥95% (HPLC)
Form
powder
Farbe
white to beige
Löslichkeit
DMSO: 2 mg/mL, clear
Lagertemp.
−20°C
Biochem./physiol. Wirkung
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumente section.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.