Wichtige Dokumente
10810
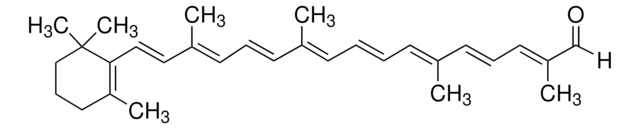
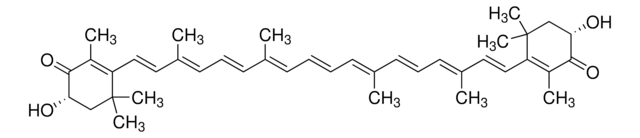
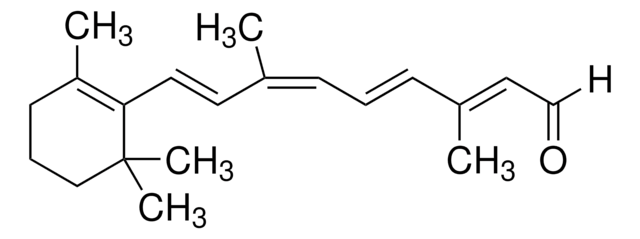
β-Apo-8′-carotinal (trans)
≥96.0% (UV)
Synonym(e):
Apocarotinal
About This Item
Empfohlene Produkte
Biologische Quelle
synthetic
Assay
≥96.0% (UV)
Form
powder
Verlust
≤0.5% loss on drying, 20 °C (HV)
mp (Schmelzpunkt)
137-141 °C
Löslichkeit
chloroform: 1 mg/mL, clear to very faintly turbid, intense red-orange
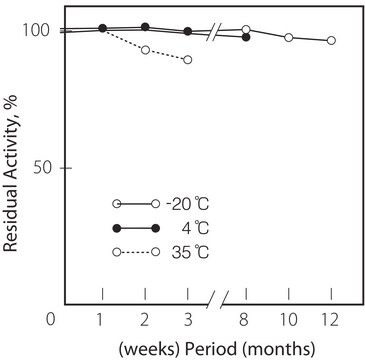
Lagertemp.
−20°C
SMILES String
[H]C(=O)\C(C)=C\C=C\C(C)=C\C=C\C=C(C)\C=C\C=C(C)\C=C\C1=C(C)CCCC1(C)C
InChI
1S/C30H40O/c1-24(13-8-9-14-25(2)16-11-18-27(4)23-31)15-10-17-26(3)20-21-29-28(5)19-12-22-30(29,6)7/h8-11,13-18,20-21,23H,12,19,22H2,1-7H3/b9-8+,15-10+,16-11+,21-20+,24-13+,25-14+,26-17+,27-18+
InChIKey
DFMMVLFMMAQXHZ-DOKBYWHISA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Anwendung
- Tackling the challenge of selective analytical clean-up of complex natural extracts: the curious case of chlorophyll removal: This study outlines a methodology for the selective analytical clean-up of complex natural extracts, which can be applied to enhance the purity and stability of trans-β-Apo-8′-carotenal, particularly useful in the context of life science manufacturing and research and development. This approach is crucial for maintaining the integrity of bioactive compounds during synthesis and storage (Bijttebier et al., 2014).
Biochem./physiol. Wirkung
Sonstige Hinweise
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 2
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.