Wichtige Dokumente
92549
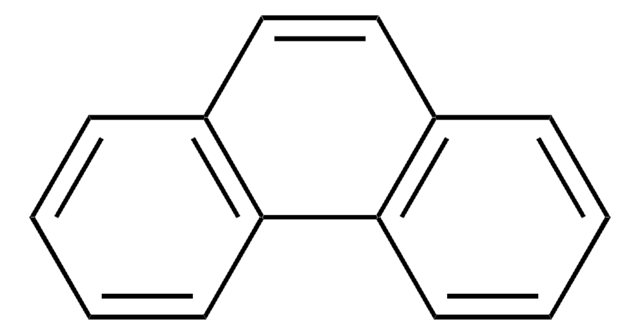
Acenaphthylin
certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
About This Item
Empfohlene Produkte
Qualität
certified reference material
TraceCERT®
Qualitätsniveau
Produktlinie
TraceCERT®
Haltbarkeit
limited shelf life, expiry date on the label
Hersteller/Markenname
Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
Methode(n)
HPLC: suitable
gas chromatography (GC): suitable
bp
280 °C (lit.)
mp (Schmelzpunkt)
78-82 °C (lit.)
89-92 °C
Dichte
0.899 g/mL at 25 °C (lit.)
Anwendung(en)
environmental
Format
neat
Lagertemp.
2-8°C
SMILES String
c1cc2C=Cc3cccc(c1)c23
InChI
1S/C12H8/c1-3-9-4-2-6-11-8-7-10(5-1)12(9)11/h1-8H
InChIKey
HXGDTGSAIMULJN-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Certified content by quantitative NMR incl. uncertainty and expiry date are given on the certificate.
Download your certificate at: http://www.sigma-aldrich.com.
Anwendung
Verpackung
Rechtliche Hinweise
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
251.6 °F - closed cup
Flammpunkt (°C)
122.0 °C - closed cup
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumente section.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Protokolle
US EPA Method 8270 (PAH only): GC Analysis of PAHs on SLB®-5ms
US EPA Method 610 describes the analysis of polynuclear aromatic hydrocarbons (commonly referred to as PAHs or PNAs) by both HPLC and GC.
GC Analysis of Polynuclear Aromatic Hydrocarbons (PAHs) in Salmon on SPB®-608 (20 m x 0.18 mm I.D., 0.18 µm) after QuEChERS Cleanup using Supel™ QuE Z-Sep, Fast GC Analysis
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.

![[(3R)-3-Hydroxydecanoyl]-L-carnitin-(N-methyl-d3) analytical standard](/deepweb/assets/sigmaaldrich/product/structures/273/425/010200db-ebc0-4746-b60c-4ac5029b9958/640/010200db-ebc0-4746-b60c-4ac5029b9958.png)