915793
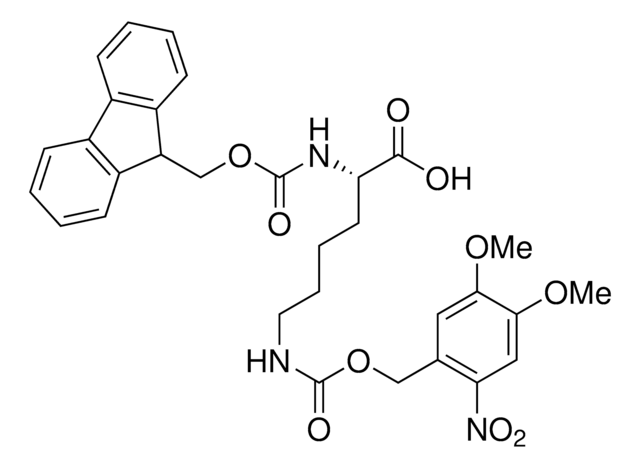
Methyl-o-nitropiperonyllysine
≥95%
Synonym(e):
N6-((1-(6-Nitrobenzo[d][1,3]dioxol-5-yl)ethoxy)carbonyl)-L-lysine, Light-triggered decaging Lys, Photo-controlled amino acid, Photocaged amino acid, Photocleavable lysine derivative, mNPK
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C16H21N3O8
CAS-Nummer:
Molekulargewicht:
383.35
MDL-Nummer:
UNSPSC-Code:
12352209
NACRES:
NA.22
Empfohlene Produkte
Anwendung
Methyl-o-nitropiperonyllysine (mNPK) trifluoroacetic acid is a photo-responsive unnatural amino acid (UAA) for spatiotemporal control of biological molecules or processes as reported by Kneuttinger et al. Irradiation with UV light decages the Lys amino acid, freeing the residue or protein for biological activity. Tools such as mNPK will find wide utility in light regulation of activity, allostery, and enzyme pathways.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Sonstige Hinweise
Light Regulation of Enzyme Allostery through Photoresponsive Unnatural Amino Acids
Precise Photoremovable Perturbation of a Virus-Host Interaction
Genetic code expansion in the mouse brain
Genetically encoded optical activation of DNA recombination in human cells
Bioorthogonal Chemical Activation of Kinases in Living Systems
Precise Photoremovable Perturbation of a Virus-Host Interaction
Genetic code expansion in the mouse brain
Genetically encoded optical activation of DNA recombination in human cells
Bioorthogonal Chemical Activation of Kinases in Living Systems
Ähnliches Produkt
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Lot/Batch Number
Leider sind derzeit keine COAs für dieses Produkt online verfügbar.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Gong Zhang et al.
ACS central science, 2(5), 325-331 (2016-06-10)
Selective manipulation of protein kinases under living conditions is highly desirable yet extremely challenging, particularly in a gain-of-function fashion. Here we employ our recently developed bioorthogonal cleavage reaction as a general strategy for intracellular activation of individual kinases. Site-specific incorporation
Sarah B Erickson et al.
Angewandte Chemie (International ed. in English), 56(15), 4234-4237 (2017-03-16)
Viruses utilize distinct binding interactions with a variety of host factors to gain entry into host cells. A chemical strategy is described to precisely perturb a specific molecular interaction between adeno-associated virus and its host cell, which can be rapidly
Hanna Engelke et al.
ACS synthetic biology, 3(10), 731-736 (2014-06-17)
Controlled manipulation of proteins and their function is important in almost all biological disciplines. Here, we demonstrate control of protein activity with light. We present two different applications-light-triggered transcription and light-triggered protease cleavage-both based on the same concept of protein
Arnaud Gautier et al.
Journal of the American Chemical Society, 133(7), 2124-2127 (2011-01-29)
We report a general strategy for creating protein kinases in mammalian cells that are poised for very rapid activation by light. By photoactivating a caged version of MEK1, we demonstrate the specific, rapid, and receptor independent activation of an artificial
Olivia S Walker et al.
Journal of the American Chemical Society, 138(3), 718-721 (2016-01-14)
Isocitrate dehydrogenase is mutated at a key active site arginine residue (Arg172 in IDH2) in many cancers, leading to the synthesis of the oncometabolite (R)-2-hydroxyglutarate (2HG). To investigate the early events following acquisition of this mutation in mammalian cells we
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.