Alle Fotos(1)
Wichtige Dokumente
512354
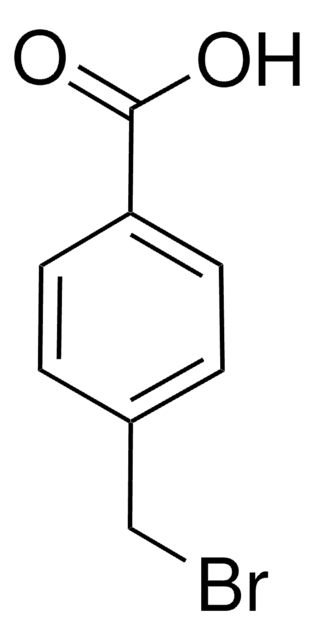
4-Benzyloxybenzoesäure
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
C6H5CH2OC6H4CO2H
CAS-Nummer:
Molekulargewicht:
228.24
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
mp (Schmelzpunkt)
189-192 °C (lit.)
Funktionelle Gruppe
carboxylic acid
phenyl
SMILES String
OC(=O)c1ccc(OCc2ccccc2)cc1
InChI
1S/C14H12O3/c15-14(16)12-6-8-13(9-7-12)17-10-11-4-2-1-3-5-11/h1-9H,10H2,(H,15,16)
InChIKey
AQSCHALQLXXKKC-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
4-Benzyloxybenzoic acid is a substituted benzoic acid that can be prepared by the benzylation of 4-hydroxybenzoic acid with benzyl bromide.
Anwendung
4-Benzyloxybenzoic acid may be used in the preparation of 1,3-phenylene bis(4-benzyloxybenzoate). It may also be used in the preparation of (-)-(2R,3R)-5,7-bis(benzyloxy)-2-[3,4,5-tris(benzyloxy)-phenyl]chroman-3-yl-(4-benzyloxy)benzoate.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral - Eye Irrit. 2
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Synthesis and characterization of achiral banana-shaped liquid crystalline molecules containing bisnaphthyl moieties.
Yang PJ and Lin HC.
Liq. Cryst., 33(5), 587-603 (2006)
Henryk Marona et al.
Acta poloniae pharmaceutica, 60(6), 477-480 (2004-04-15)
A series of alkanolamides have been tested for anticonvulsant activity in the maximal electroshock seizure (MES) and subcutaneous pentylenetetrazole seizure treshold (ScMet) assays and for neurotoxicity (TOX) in rodents. Most interesting were the anticonvulsant results of 2N-methylaminoethanol derivative II, which
Kumi Osanai et al.
Tetrahedron, 63(32), 7565-7570 (2007-08-06)
The total and semi syntheses of (2R, 3R)-epigallocatechin-3-O-(4-hydroxybenzoate), a novel catechin from Cistus salvifolius, was accomplished. The proteasome inhibition and cytotoxic activities of the synthetic compound and its acetyl derivative were studied and compared with (2R, 3R)-epigallocatechin-3-gallate (EGCG), the active
Semi-synthesis and proteasome inhibition of D-ring deoxy analogs of (-)-epigallocatechin gallate (EGCG), the active ingredient of green tea extract.
Huo C, et al.
Canadian Journal of Chemistry, 86(6), 495-502 (2008)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.