436798
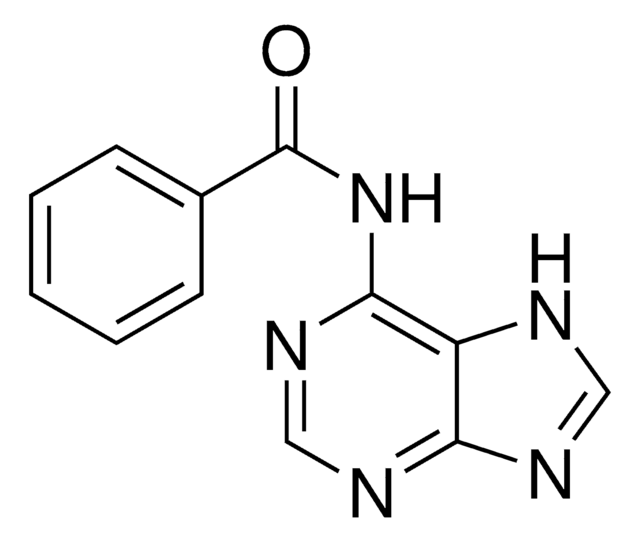
N4-Benzoylcytosin
98%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C11H9N3O2
CAS-Nummer:
Molekulargewicht:
215.21
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
mp (Schmelzpunkt)
>300 °C (dec.) (lit.)
Funktionelle Gruppe
amide
phenyl
SMILES String
O=C1NC=CC(NC(=O)c2ccccc2)=N1
InChI
1S/C11H9N3O2/c15-10(8-4-2-1-3-5-8)13-9-6-7-12-11(16)14-9/h1-7H,(H2,12,13,14,15,16)
InChIKey
XBDUZBHKKUFFRH-UHFFFAOYSA-N
Allgemeine Beschreibung
N4-Benzoylcytosine is an amide and its anti-microbial activity against pathogenic microorganisms has been studied using the Disk Diffusion and the Pour Plate method. It can be synthesized via the condensation of benzoyl chloride with cytosine.
Anwendung
N4-Benzoylcytosine may be employed for the following syntheses:
- 3′-C-ethynyl and 3′-C-(1,4-disubstituted-1,2,3-triazolo) double-headed pyranonucleosides
- 2′-C-methyl-4′-thiocytidine, via the Pummerer reaction
- 2′-fluorinated L-nucleoside analogs
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Christos Kiritsis et al.
Medicinal chemistry (Shariqah (United Arab Emirates)), 8(3), 320-329 (2012-04-26)
A novel series of 3'-C-ethynyl and 3'-C-(1,4-disubstituted-1,2,3-triazolo) double-headed pyranonucleosides has been designed and synthesized. Reaction of 3-keto glucoside 1 with ethynyl magnesium bromide gave the desired precursor 3-C-ethynyl-1,2:5,6-di-O-isopropylidene-α-D-allofuranose (2). Hydrolysis followed by acetylation led to the 1,2,4,6-tetra-O-acetyl-3-C-ethynyl-β-D-allopyranose (3). Compound 3
Daisuke Kaga et al.
Nucleosides, nucleotides & nucleic acids, 24(10-12), 1789-1800 (2006-01-28)
The synthesis of 2'-C-methyl-4'-thiocytidine (16) is described. Since the 2'-keto-4'-thiocytidine derivative 2beta unexpectedly isomerized to 2alpha and the methylation of 2beta proceeded predominantly from the less hindered alpha-face to give 7, the desired product 16 was synthesized via the Pummerer
Antimicrobial activity of amide, N4-benzoylcytosine.
Jagessar RC and Gomathinayagam S.
Journal of Pharmacy and Clinical Sciences, 1, 12-19 (2011)
K Lee et al.
Journal of medicinal chemistry, 42(7), 1320-1328 (1999-04-10)
The synthesis of L-nucleoside analogues containing 2'-vinylic fluoride was accomplished by direct condensation method, and their anti-HIV and anti-HBV activities were evaluated in vitro. The key intermediate 8, the sugar moiety of our target compounds, was prepared from 1,2-O-isopropylidene-L-glyceraldehyde via
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.