Alle Fotos(2)
Wichtige Dokumente
378895
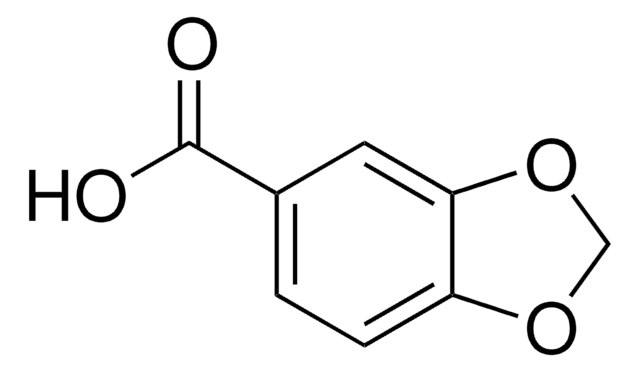
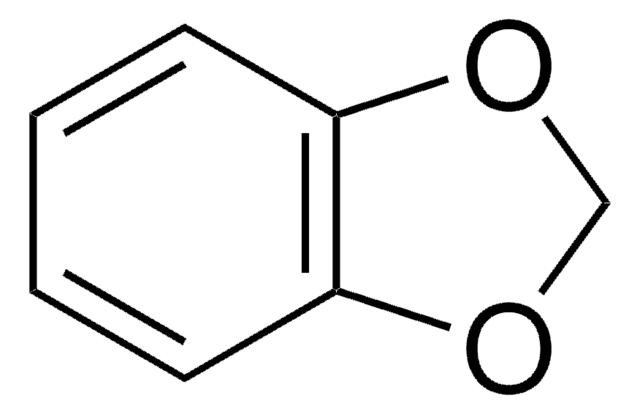
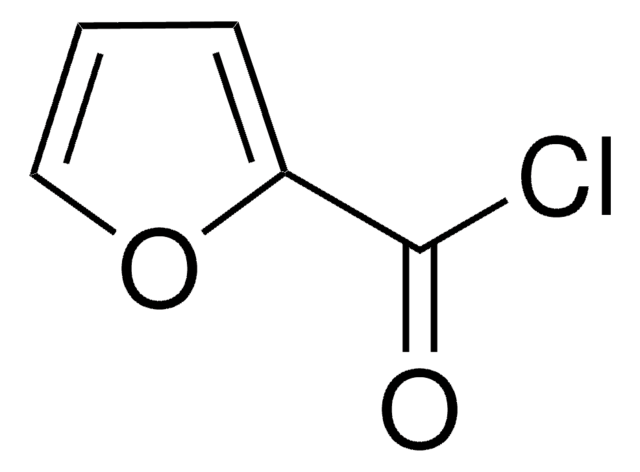
Piperonyloylchlorid
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C8H5ClO3
CAS-Nummer:
Molekulargewicht:
184.58
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
99%
bp
155 °C/25 mmHg (lit.)
mp (Schmelzpunkt)
78-79 °C (lit.)
Funktionelle Gruppe
acyl chloride
SMILES String
ClC(=O)c1ccc2OCOc2c1
InChI
1S/C8H5ClO3/c9-8(10)5-1-2-6-7(3-5)12-4-11-6/h1-3H,4H2
InChIKey
ZRSGZIMDIHBXIN-UHFFFAOYSA-N
Allgemeine Beschreibung
Piperonyloyl chloride is an acyl halide. It participates in the preparation of starting reagent (N-acyl indole), required for the synthesis of pyrrolophenanthridone alkaloids. Kinetic study of the solvolysis of piperonyloyl chloride in various pure and binary solvent mixtures has been proposed. Solvolysis reaction has been reported to proceed via electron-rich acyl transfer mechanism.
Anwendung
Piperonyloyl chloride is suitable for use in a kinetic study to evaluate the solvolysis rate constants of piperonyloyl chloride in 27 different solvents. It may be used in the synthesis of the following compounds:
- 2-phenylbenzimidazoles
- (Z)-3-hydroxy-1-(5-methoxy-2,2-dimethyl-2H-chromen-6-yl)-3-phenylprop-2-en-1-one
- pongapinone A
- 2-((1-(2-(N-(4-chlorophenyl)benzo[d][1,3]dioxole-5-carboxamido)ethyl)piperidin-4-yl)oxy)acetic acid phosphoric acid salt, inhibitor of platelet aggregation
- justicidin B, the piscicidal components of Justicia Hayatai var. decumbens
- piperazine derivatives
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Eye Dam. 1 - Skin Corr. 1B
Lagerklassenschlüssel
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Pelletier SW.
Alkaloids: Chemical and Biological Perspectives, Volume 14, 14, 441-442 (2000)
Studies on the Piscicidal Components of Justicia Hayatai var. decumbens.
OHTA K, et al.
Agricultural and Biological Chemistry, 33(4), 610-614 (1969)
Serena Scapecchi et al.
Bioorganic & medicinal chemistry, 12(1), 71-85 (2003-12-31)
Structure-activity relationships on two novel potent cognition enhancing drugs, unifiram (DM232, 1) and sunifiram (DM235, 2), are reported. Although none of the compounds synthesised reached the potency of the parent drugs, some fairly active compounds have been identified that may
Concise Synthetic Approaches to Naturally Occurring ?-Hydroxypyranochalcones: First Total Synthesis of Purpurenone, Its Derivative, and Praecansone B.
Wang X, et al.
Bull. Korean Chem. Soc., 33(8), 2647-2650 (2012)
Hachemi Kadri et al.
Journal of enzyme inhibition and medicinal chemistry, 23(5), 641-647 (2008-09-30)
A new series of fluorinated and non-fluorinated 2-phenylbenzimidazoles bearing oxygenated substituents on the phenyl ring has been synthesized. Synthesis of the new series was based on our previous discovery of 2-(3,4-dimethoxyphenyl)-5-fluorobenzothiazole (PMX 610) as a potent and selective antitumour agent
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.