371475
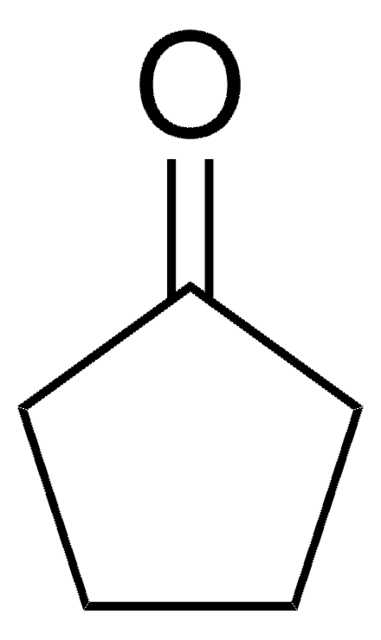
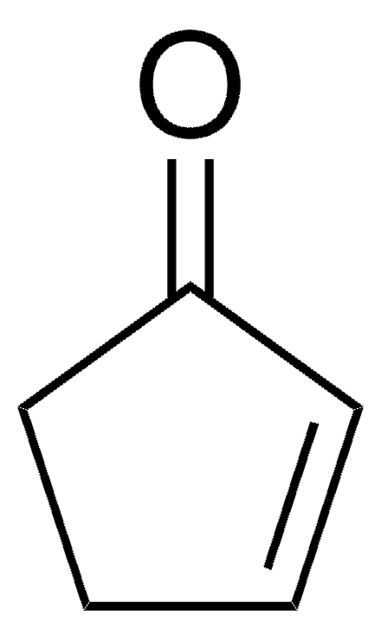
2,2-Dimethylcyclopentanon
96%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Lineare Formel:
(CH3)2C5H6(=O)
CAS-Nummer:
Molekulargewicht:
112.17
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
96%
Brechungsindex
n20/D 1.433 (lit.)
bp
143-145 °C (lit.)
Dichte
0.894 g/mL at 25 °C (lit.)
Funktionelle Gruppe
ketone
SMILES String
CC1(C)CCCC1=O
InChI
1S/C7H12O/c1-7(2)5-3-4-6(7)8/h3-5H2,1-2H3
InChIKey
FTGZMZBYOHMEPS-UHFFFAOYSA-N
Allgemeine Beschreibung
2,2-Dimethylcyclopentanone is a ketone. Synthesis of various C-2 substituted vitamin D derivatives with a 2,2-dimethylcyclopentanone unit in side chains has been reported. Regioselective synthesis of 2,2-dimethylcyclopentanone, via its enolate precursor regioselectively obtained using the 2-pyrrolidone magnesium salt, has been reported.
Anwendung
2,2-Dimethylcyclopentanone enolate may be used as starting reagent in the enantioselective synthesis of chiral phosphines belonging to the P-aryl-2-phosphabicyclo[3.3.0]octane family (PBO). It may be used in the synthesis of:
- 2,6,6-trimethyl-2-azaspiro[4.4]nonane-1,3-dione, a spirosuccinimide moiety of asperparaline A
- novel spiropentanopyrrolizidine oxime alkaloids, namely 2′,3′,5′,6′,7′,7a′-hexahydro-2,2-dimethylspirocyclopentane-1
- δ,δ-dimethyl-δ-valerolactone, via Baeyer-Villiger oxidation
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Regioselective Synthesis of 2, 2-Dimethylcyclopentanone Using 2-Pyrrolidone Magnesium Salt as Electrogenerated Base.
Bonafoux D, et al.
Synthetic Communications, 28(1), 93-98 (1998)
Hiroshi Saito et al.
The Journal of steroid biochemistry and molecular biology, 136, 3-8 (2013-02-19)
Up to the present, numerous vitamin D derivatives have been synthesized, but most of them have straight side chains, and there are few publications described about in vitro and in vivo evaluations on bone by vitamin D derivatives. In our
S Tanimori et al.
Bioscience, biotechnology, and biochemistry, 64(8), 1758-1760 (2000-09-19)
2,6,6-Trimethyl-2-azaspiro[4.4]nonane-1,3-dione (9), a spirosuccinimide moiety of asperparaline A (1), was synthesized by starting from 2,2-dimethylcyclopentanone (4) via trinitrile 6 in five steps in a moderate yield. This conversion establishes a model study for synthesis of the spirosuccinimide moiety of asperparaline
Edwin Vedejs et al.
Journal of the American Chemical Society, 125(14), 4166-4173 (2003-04-03)
A new class of chiral phosphines belonging to the P-aryl-2-phosphabicyclo[3.3.0]octane family (PBO) has been prepared by enantioselective synthesis starting from lactate esters and 2,2-dimethylcyclopentanone enolate 5. A selective enolate alkylation method has been developed for preparation of 9 and 10
Synthesis of pyrrolizidine oximes 222 and 236: Novel alkaloids of a dendrobatid poison frog.
Rutchinson KD, et al.
Tetrahedron, 50(21), 6129-6136 (1994)
Artikel
The Baeyer-Villiger oxidation is the oxidative cleavage of a carbon-carbon bond adjacent to a carbonyl, which converts the ketones to esters and the cyclic ketones to lactones.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.