P4234
Pyranose Oxidase from Coriolus sp.
recombinant, expressed in E. coli, ≥2.7 units/mg solid
Synonyme(s) :
Pyranose: Oxygen 2-Oxidoreductase
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Numéro CAS:
Numéro MDL:
Code UNSPSC :
12352204
Nomenclature NACRES :
NA.54
Produits recommandés
Produit recombinant
expressed in E. coli
Forme
powder
Activité spécifique
≥2.7 units/mg solid
Conditions d'expédition
wet ice
Température de stockage
−20°C
Description générale
Pyranose oxidase (P2O), a homotetrameric protein consists of a covalently bound flavin adenine dinucleotide (FAD). It is seen mostly among wood-degrading basidiomycetes.
Application
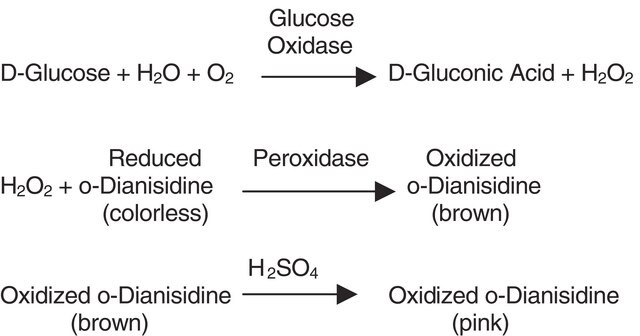
Pyranose Oxidase from Coriolus sp. has been used in the enzymatic oxidation of D-glucose (DG). It has also been used as a component in oxygen scavenging system (OSS) to increase the lifetime of the fluorophores.
Actions biochimiques/physiologiques
Pyranose oxidase (P2O) can be used in clinical chemistry to determine 1,5-anhydro-d-glucitol marker, used for glycemic control in diabetes patients.
Pyranose oxidase (P2O) catalyzes the oxidation of aldopyranoses at position C-2 to yield the corresponding 2-ketoaldoses. The in vivo substrates of P2O are thought to be D-glucose, D-galactose, and D-xylose. They are oxidized to 2-keto-D-glucose (D-arabino-hexos-2-ulose, 2-dehydro-D-glucose), 2-keto-D-galactose (D-lyxo-hexos-2-ulose, 2-dehydro-D-galactose), and 2-keto-D-xylose (D-threopentos-2-ulose, 2-dehydro-D-xylose), respectively. Pyranose oxidase has significant activity with carbohydrates such as, L-sorbose, D-glucono-1,5-lactone, and D-allose. When pyranose oxidase catalyzes the oxidation of aldopyranoses, electrons are transferred to molecular oxygen which results in the formation of hydrogen peroxide.
Définition de l'unité
One unit produces 1.0 μmol of hydrogen peroxide per minute at 37 °C, pH 7.0.
Autres remarques
Contains glutamate
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Resp. Sens. 1
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 1
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
The influence of temperature on bioconversion of D-glucose into 2-keto-D-glucose by Pyranose oxidase
Ene MD, et al.
UPB Scientific Bulletin, Series B: Chemistry and Materials Science, 2 (2014)
Oliver Spadiut et al.
Microbial cell factories, 9, 14-14 (2010-03-11)
The heterologous production of the industrially relevant fungal enzyme pyranose 2-oxidase in the prokaryotic host E. coli was investigated using 3 different expression systems, i.e. the well-studied T7 RNA polymerase based pET21d+, the L-arabinose inducible pBAD and the pCOLD system.
Antti Lignell et al.
Nature communications, 8(1), 1830-1830 (2017-12-01)
The neural crest is an embryonic population of multipotent stem cells that form numerous defining features of vertebrates. Due to lack of reliable techniques to perform transcriptional profiling in intact tissues, it remains controversial whether the neural crest is a
Warintra Pitsawong et al.
The Journal of biological chemistry, 285(13), 9697-9705 (2010-01-22)
Pyranose 2-oxidase (P2O) catalyzes the oxidation by O(2) of d-glucose and several aldopyranoses to yield the 2-ketoaldoses and H(2)O(2). Based on crystal structures, in one rotamer conformation, the threonine hydroxyl of Thr(169) forms H-bonds to the flavin-N5/O4 locus, whereas, in
Tien-Chye Tan et al.
Journal of molecular biology, 402(3), 578-594 (2010-08-17)
Flavoenzymes perform a wide range of redox reactions in nature, and a subclass of flavoenzymes carry covalently bound cofactor. The enzyme-flavin bond helps to increase the flavin's redox potential to facilitate substrate oxidation in several oxidases. The formation of the
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique