78825
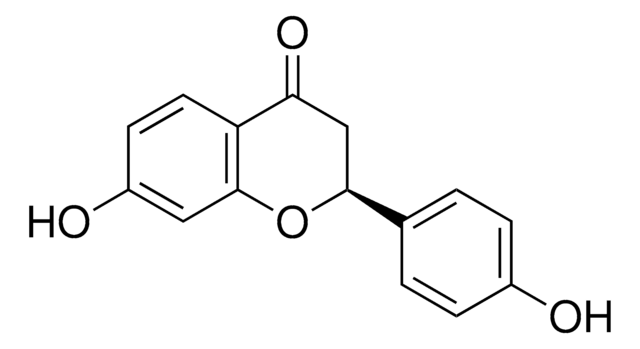
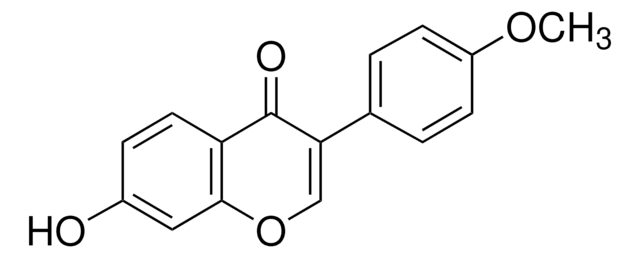
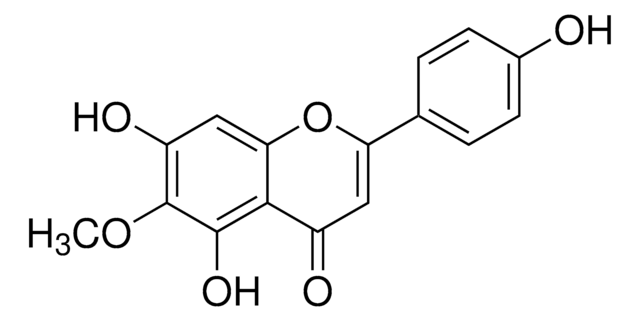
Liquiritigenin
≥97.0% (HPLC)
Synonyme(s) :
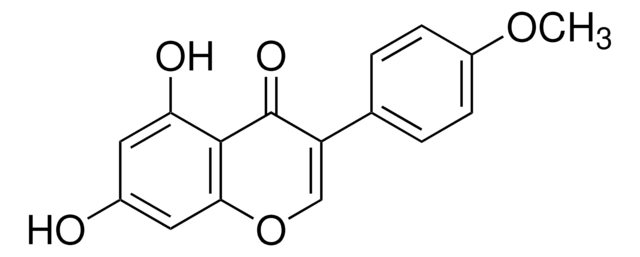
7,4′-Dihydroxyflavanone, 7-Hydroxy-2-(4-hydroxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one
About This Item
Produits recommandés
Niveau de qualité
Essai
≥97.0% (HPLC)
Forme
powder or crystals
Impuretés
≤7% water
Application(s)
metabolomics
vitamins, nutraceuticals, and natural products
Chaîne SMILES
Oc1ccc(cc1)[C@@H]2CC(=O)c3ccc(O)cc3O2
InChI
1S/C15H12O4/c16-10-3-1-9(2-4-10)14-8-13(18)12-6-5-11(17)7-15(12)19-14/h1-7,14,16-17H,8H2/t14-/m0/s1
Clé InChI
FURUXTVZLHCCNA-AWEZNQCLSA-N
Description générale
Application
- to study its inhibitory effect on tumor metastasis in the treatment of colorectal cancer
- as a reference standard for ultra-performance liquid chromatography (UPLC) of Chaihu-Shugan-San (CSS) extract
- as a potential antiviral drug against hepatitis C virus (HCV) infection
Actions biochimiques/physiologiques
Conditionnement
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique