T6508
Acide trifluoroacétique
ReagentPlus®, 99%
Synonyme(s) :
TFA
About This Item
Produits recommandés
Densité de vapeur
3.9 (vs air)
Niveau de qualité
Pression de vapeur
97.5 mmHg ( 20 °C)
Gamme de produits
ReagentPlus®
Pureté
99%
Forme
liquid
Impuretés
≤0.05% water
Indice de réfraction
n20/D 1.3 (lit.)
pH
1 (10 g/L)
Point d'ébullition
72.4 °C (lit.)
Pf
−15.4 °C (lit.)
Solubilité
ethanol: soluble 0.33 mL/mL
Densité
1.489 g/mL at 20 °C (lit.)
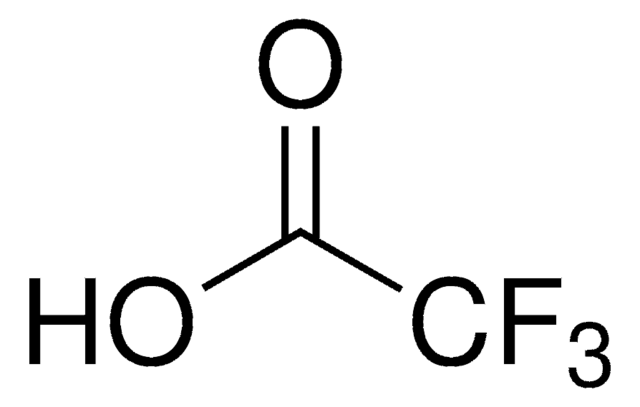
Chaîne SMILES
OC(C(F)(F)F)=O
InChI
1S/C2HF3O2/c3-2(4,5)1(6)7/h(H,6,7)
Clé InChI
DTQVDTLACAAQTR-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- pour cliver les groupements protecteurs de l'azote et de l'oxygène tels que les groupements N-Boc, N-benzyloxyméthyl, benzyl éther, p-méthoxybenzyl éther, t-butyl éther, t-butyloxyméthyl éther, triphénylméthyl éther et diméthyl acétal.
- dans les réactions d'oxydation de Baeyer–Villiger en association avec le percarbonate de sodium.·
- dans la trifluorométhylation des liaisons C-H des arènes.
Le TFA peut aussi être employé :
- comme solvant dans les réactions de cyclisation par transfert d'atome et les procédés de formation de polymères.
- comme catalyseur dans la synthèse de ε-caprolactame par réarrangement de Beckmann de l'oxime de cyclohexanone dans des solvants aprotiques.
Conditionnement
Informations légales
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Acute Tox. 4 Inhalation - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1A
Code de la classe de stockage
8A - Combustible corrosive hazardous materials
Classe de danger pour l'eau (WGK)
WGK 2
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Contenu apparenté
Fmoc resin cleavage and deprotection follows the difficult task of detaching the peptide from the resin support and removing all the side-chain protecting groups of the amino acid residues to yield the desired peptide.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique