17849
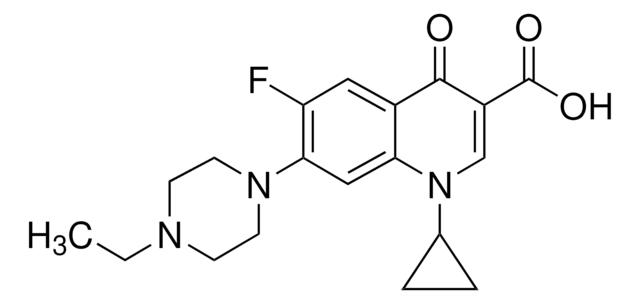
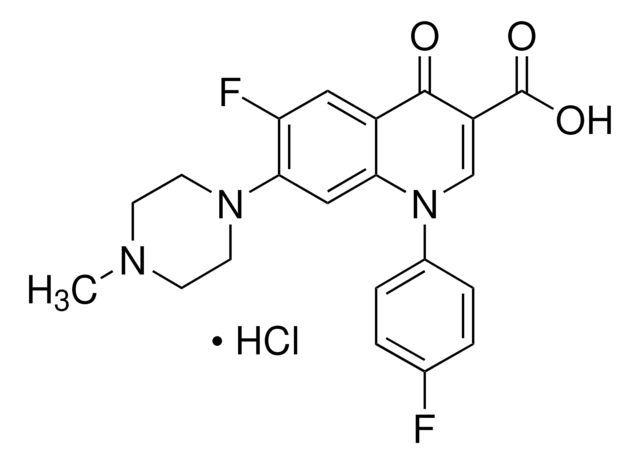
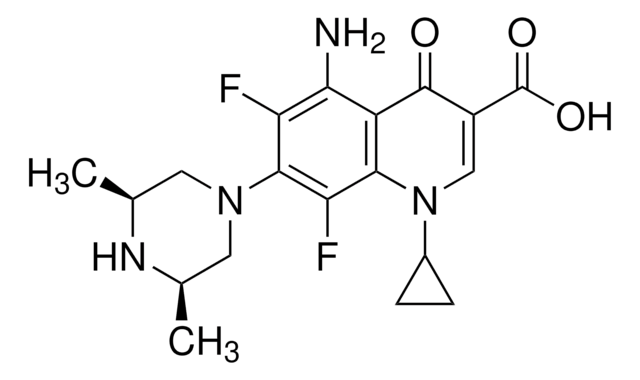
Enrofloxacin
≥99.0%
Synonyme(s) :
Baytril
About This Item
Produits recommandés
Niveau de qualité
Pureté
≥99.0%
99.0-101.0% (dried substance)
Forme
powder or crystals
Spectre d'activité de l'antibiotique
Gram-negative bacteria
Gram-positive bacteria
Mode d’action
DNA synthesis | interferes
enzyme | inhibits
Chaîne SMILES
CCN1CCN(CC1)c2cc3N(C=C(C(O)=O)C(=O)c3cc2F)C4CC4
InChI
1S/C19H22FN3O3/c1-2-21-5-7-22(8-6-21)17-10-16-13(9-15(17)20)18(24)14(19(25)26)11-23(16)12-3-4-12/h9-12H,2-8H2,1H3,(H,25,26)
Clé InChI
SPFYMRJSYKOXGV-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- to investigate the pathological mechanisms resulting from the toxicity of fluoroquinolones in mammalian cells
- to prepare a standard solution with 50% acetonitrile in a study to analyze illegal fish drugs used in aquaculture by employing surface-enhanced Raman spectroscopy (SERS)
- as an analyte in a chemiluminescence reagent system
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Oral - Resp. Sens. 1 - Skin Sens. 1
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique