810600P
Avanti
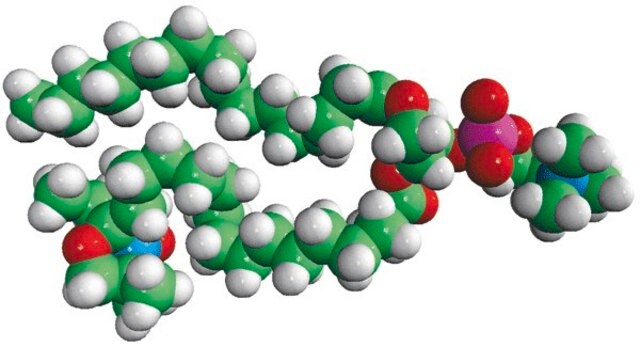
16:0-12 Doxyl PC
Avanti Research™ - A Croda Brand 810600P, powder
Synonyme(s) :
1-palmitoyl-2-stearoyl-(12-doxyl)-sn-glycero-3-phosphocholine
About This Item
Produits recommandés
Essai
>99% (TLC)
Forme
powder
Conditionnement
pkg of 1 × 1 mg (810600P-1mg)
Fabricant/nom de marque
Avanti Research™ - A Croda Brand 810600P
Type de lipide
ESR probes
phospholipids
Conditions d'expédition
dry ice
Température de stockage
−20°C
Description générale
Application
- as a lipophilic collisional quencher to prepare liposomes used in lipophilic quenching experiments
- to prepare liposomes used in nitroxide quenching experiments to examine the location of each NBD (7-nitrobenz-2-oxa-1,3-diazole) probe in mutants
- as a component in POPC or 1:1 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC)/1-palmitoyl-2-oleoyl-sn-glycero-3-phospho-rac-1-glycerol (POPG) mixtures to prepare unlabelled large unilamellar liposomes
Actions biochimiques/physiologiques
Conditionnement
Notes préparatoires
Informations légales
Souvent commandé avec ce produit
Code de la classe de stockage
11 - Combustible Solids
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
Si vous avez besoin d'assistance, veuillez contacter Service Clients
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique