377198
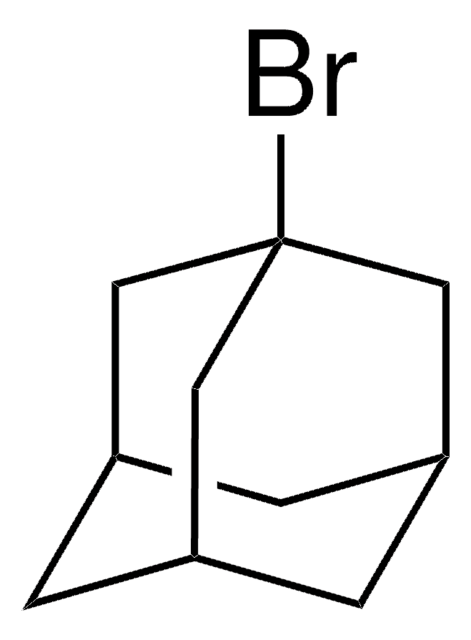
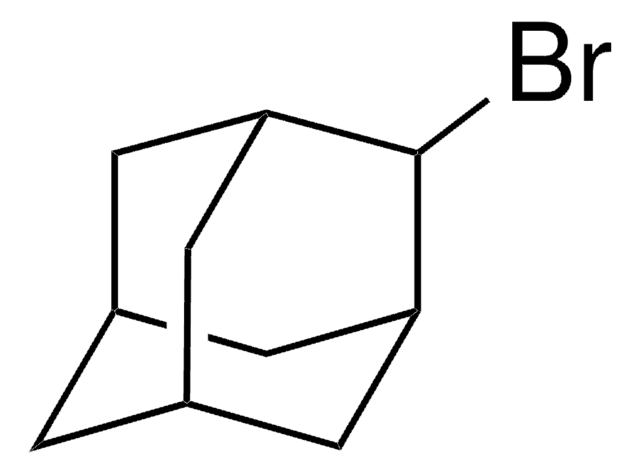
1-Iodoadamantane
98%
Synonyme(s) :
1-Adamantyl iodide, 1-Iodotricyclo[3.3.1.13,7]decane, Adamantyl iodide
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
C10H15I
Numéro CAS:
Poids moléculaire :
262.13
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Essai
98%
Forme
solid
Pf
75-76 °C (lit.)
Groupe fonctionnel
iodo
Chaîne SMILES
IC12C[C@H]3C[C@H](C[C@H](C3)C1)C2
InChI
1S/C10H15I/c11-10-4-7-1-8(5-10)3-9(2-7)6-10/h7-9H,1-6H2/t7-,8+,9-,10-
Clé InChI
PXVOATXCSSPUEM-CHIWXEEVSA-N
Description générale
1-Iodoadamantane is a haloadamantane. Voltammetric reduction of 1-iodoadamantane at a silver cathode in tetrahydrofuran (THF) and acetonitrile (ACN) is reported to involve a single electron forming a mixture of monomeric and dimeric products. The photoinduced reaction of 1-iodoadamantane in DMSO is reported to afford substitution products on C3, C6, and C8, 1-adamantanol, 1-adamantyl 2-naphthyl ether, and adamantine. The photostimulated reaction of the phthalimide anion with 1-iodoadamantane is reported to yield 3-(1-adamantyl) phthalimide and 4-(1-adamantyl) phthalimide, along with the reduction product adamantane. 1-Iodoadamantane is reported to undergoe photostimulated reaction with the enolate anion of acetone, acetophenone and propiophenone to give admantane and the substitution products.
Application
1-Iodoadamantane is suitable reagent used to evaluate the rate constants for the reduction of haloadamantanes by SmI2 in presence of hexamethylphosphoramide (HMPA) and H2O by GC/MS-analyzed method. It may be used as iodine atom donor for probing the intermediacy of radical to investigate the chemistry of highly reactive, strained systems such as propellane. It may be used as starting reagent in the synthesis of N-(1-adamantyl)acetamide via nucleophilic substitution. It may be employed in the free-radical carbonylation reactions with alkenes.
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Photostimulated reaction of 1-iodoadamantane with carbanionic nucleophiles in DMSO by the SRN1 mechanism.
Borosky GL, et al.
The Journal of Organic Chemistry, 55(12), 3705-3707 (1990)
Juan E Argüello et al.
The Journal of organic chemistry, 68(6), 2362-2368 (2003-03-15)
The fluorescent excited state of the 2-naphthoxide ion (1) is quenched by aliphatic and aromatic halides according to an electron-transfer mechanism, with generation of the corresponding alkyl and aryl radicals by a concerted or consecutive C-X bond fragmentation reaction. Whereas
Dimerization of Cubene. 1-Iodoadamantane as a Probe for Radical Intermediates.
Lukin K and Eaton PE.
Journal of the American Chemical Society, 117(29), 7652-7656 (1995)
Rate study of haloadamantane reduction by samarium diiodide.
Lin T-Y, et al.
J. Chin. Chem. Soc., 49(6), 969-973 (2002)
Christopher A Paddon et al.
Ultrasonics sonochemistry, 14(5), 502-508 (2007-01-17)
The combination of ultrasound and electrochemistry -sonoelectrochemistry can produce a variety of effects within an electrochemical system including enhanced mass transport, in situ cleaning of an electrode surface, diminution of the diffusion layer, and possible induction of new reactions by
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique