N9284
Nitroreductase from Escherichia coli
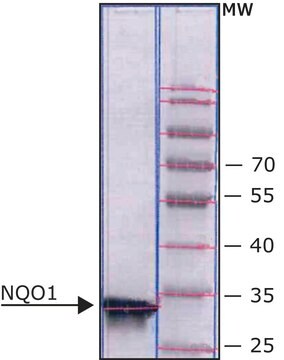
≥90% (SDS-PAGE), recombinant, expressed in E. coli
Synonym(s):
NTRA
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Recommended Products
recombinant
expressed in E. coli
Quality Level
Assay
≥90% (SDS-PAGE)
form
lyophilized powder
specific activity
≥100 units/mL
mol wt
monomer 24000
greener alternative product characteristics
Waste Prevention
Safer Solvents and Auxiliaries
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
UniProt accession no.
greener alternative category
shipped in
wet ice
storage temp.
−20°C
Gene Information
Escherichia coli K12 ... nfsB(945483)
General description
Nitroreductase is a flavoprotein and is encoded by the NfsB gene. It comprises a dimer, with 217 amino acids and active site in each subunit. The structure has FMN and the substrate bound to the enzyme.
Application
Nitroreductase from Escherichia coli has been used in the conjugation generation with pig liver esterase (PLE). It has also been used in chemiluminescence response studies with probes HyCL-3 and HyCL-4-AM in rat liver microsomes.
Biochem/physiol Actions
Nitroreductase (NTR) catalyzes the reduction of nitroaromatic substrates and quinones. The mutant F124K of NTR is useful in cancer therapy and improves sensitization of drug CB1954.
Nitroreductase increases the sensitivity of organisms to nitro-containing drugs such as metronidazole by converting the nitro group to a cytotoxic nitro radical.
Nitroreductases can play a crucial role in redox systems via NADPH or NADH as a reductant.
Shows ability to reduce quinines. Enzyme for activating prodrugs in antibody directed enzyme prodrug therapy.
Physical properties
Lyophilized powder containing PBS. Does not contain BSA as excipient
Unit Definition
One unit will reduce one μmole of Cytochrome C per minute in the presence of Menadione and NADH at pH 7.4 at 37 °C.
Preparation Note
Produced using animal component-free materials.
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Dual enzyme-responsive ?turn-on? fluorescence sensing systems based on in situ formation of 7-hydroxy-2-iminocoumarin scaffolds
Debieu, S and Romieu A
Organic & Biomolecular Chemistry, 13(41), 10348-10361 (2015)
Chih-Chen Chen et al.
Food chemistry, 135(4), 2708-2713 (2012-09-18)
Nitroreductases (Nrs) play important roles in redox system via NADPH or NADH as a reductant. A TcNr cDNA encoding a putative Nr was cloned from Taiwanofungus camphorata. A 3-D structural model of the TcNr has been created based on the
Mansooreh Jaberipour et al.
Biochemical pharmacology, 79(2), 102-111 (2009-08-12)
Prodrug activation gene therapy for cancer involves expressing prodrug-activating enzymes in tumour cells, so they can be selectively killed by systemically administered prodrug. For example, Escherichia colinfsB nitroreductase (E.C. 1.6.99.7)(NTR), sensitises cells to the prodrug CB1954 (5-[aziridin-1-yl]-2,4-dinitrobenzamide), which it converts
Bharat Bhushan et al.
Biochemical and biophysical research communications, 322(1), 271-276 (2004-08-18)
Previously, we reported that a salicylate 1-monooxygenase from Pseudomonas sp. ATCC 29352 biotransformed CL-20 (2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaaza-isowurtzitane) (C(6)H(6)N(12)O(12)) and produced a key metabolite with mol. wt. 346 Da corresponding to an empirical formula of C(6)H(6)N(10)O(8) which spontaneously decomposed in aqueous medium to
M J Lemmon et al.
Gene therapy, 4(8), 791-796 (1997-08-01)
A fundamental obstacle in gene therapy for cancer treatment is the specific delivery of an anticancer gene product to a solid tumor. Although several strategies exist to control gene expression once a vector is directly introduced into a tumor, as
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![Nitrate Reductase (NAD[P]H) from Aspergillus niger lyophilized powder, ≥300 units/g solid](/deepweb/assets/sigmaaldrich/product/images/309/282/2a67ae4d-ca55-4f0b-96ec-34748ff8a21e/640/2a67ae4d-ca55-4f0b-96ec-34748ff8a21e.jpg)