SMB01361
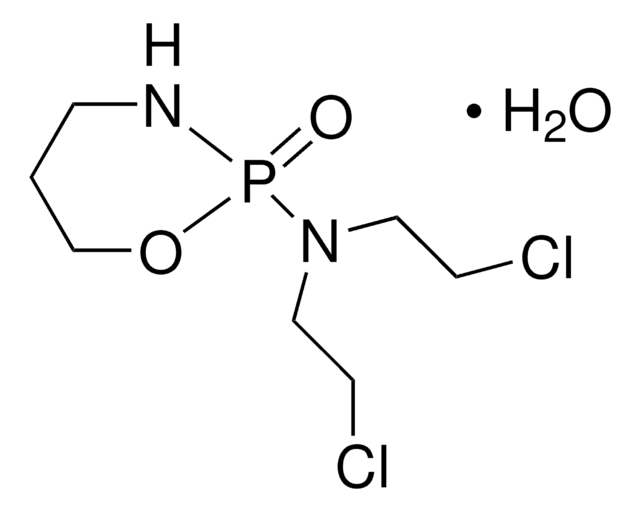
Phosphoramide mustard cyclohexylamine
≥95% (HPLC)
Synonym(s):
N,N-Bis(2-Chloroethyl)phosphorodiamidic acid, cyclohexylamine, Friedman acid, cyclohexylamine, PAM cyclohexylamine, PM cyclohexylamine, PMC, Phosphamide mustard, Cyclohexylamine, Phosphorodiamidic mustard, Cyclohexylamine
About This Item
Recommended Products
Quality Level
Assay
≥95% (HPLC)
form
solid
storage temp.
-10 to -25°C
SMILES string
ClCCN(P(N)(O)=O)CCCl.NC1CCCCC1
InChI
1S/C6H13N.C4H11Cl2N2O2P/c7-6-4-2-1-3-5-6;5-1-3-8(4-2-6)11(7,9)10/h6H,1-5,7H2;1-4H2,(H3,7,9,10)
InChI key
BGTIPRUDEMNRIP-UHFFFAOYSA-N
General description
Application
Biochem/physiol Actions
Features and Benefits
- High quality compound suitable for multiple research applications
- Compatible with a wide variety of chromatographic and spectrometry techniques
Other Notes
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service