742430
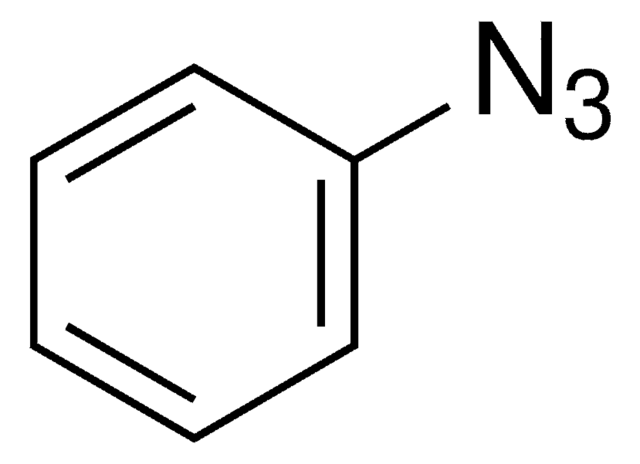
Benzyl azide solution
~0.5 M in dichloromethane, ≥95.0% (HPLC)
Synonym(s):
α-Azidotoluene solution, (Azidomethyl)benzene solution
About This Item
Recommended Products
Quality Level
Assay
≥95.0% (HPLC)
form
solution
concentration
~0.5 M in dichloromethane
impurities
≤2.0% water
storage temp.
2-8°C
SMILES string
[N-]=[N+]=NCc1ccccc1
InChI
1S/C7H7N3/c8-10-9-6-7-4-2-1-3-5-7/h1-5H,6H2
InChI key
UDLLFLQFQMACJB-UHFFFAOYSA-N
General description
Application
- Synthesis of Estrone Heterodimers and Evaluation of Their In Vitro Antiproliferative Activity.: This research investigates the synthesis of estrone heterodimers using benzyl azide as a key reagent, demonstrating significant antiproliferative activity, thus highlighting its potential in pharmaceutical intermediate synthesis (Bózsity N et al., 2024).
- Fluoride Abstraction Induced by Tris(pentafluoroethyl)difluorophosphorane: A Convenient Way to Synthesize Cationic N-Heterocyclic Carbene- and Cyclic (Alkyl)(amino)carbene-Ligated Copper Alkyne and Arene Complexes.: This paper describes a method for synthesizing copper alkyne and arene complexes using benzyl azide, emphasizing its role in innovative click chemistry applications (Riethmann M et al., 2024).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Carc. 2 - Eye Irrit. 2 - Skin Irrit. 2 - STOT RE 2 - STOT SE 3
Target Organs
Central nervous system
Storage Class Code
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service