685828
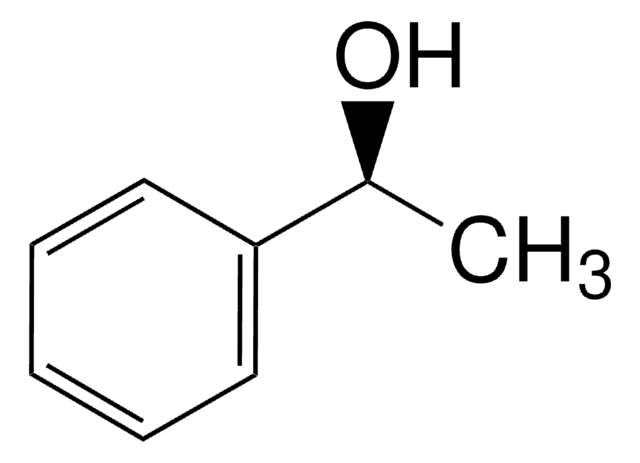
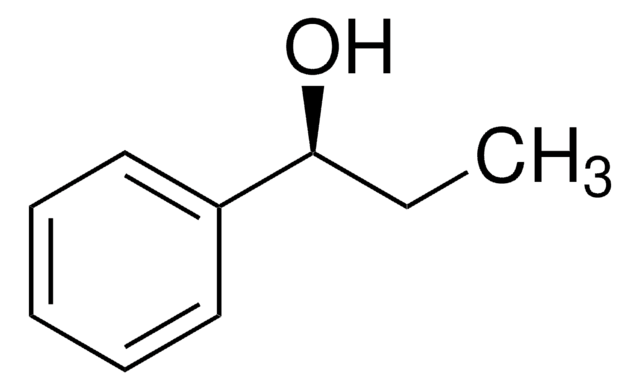
(R)-1-Phenylethanol
97%
Synonym(s):
(+)-α-Methylbenzyl alcohol, (+)-α-Phenethyl alcohol, (+)-α-Phenylethanol, (+)-1-Phenylethanol, (+)-1-Phenylethyl alcohol, (+)-sec-Phenethyl alcohol
About This Item
Recommended Products
Quality Level
Assay
97%
form
liquid
optical activity
[α]22/D +44.0°, c = 5%
refractive index
n20/D 1.528
density
0.9986 g/mL at 25 °C
functional group
hydroxyl
phenyl
SMILES string
C[C@@H](O)c1ccccc1
InChI
1S/C8H10O/c1-7(9)8-5-3-2-4-6-8/h2-7,9H,1H3/t7-/m1/s1
InChI key
WAPNOHKVXSQRPX-SSDOTTSWSA-N
General description
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
185.0 °F - closed cup
Flash Point(C)
85 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Chromatograms
suitable for GCsuitable for GCGlobal Trade Item Number
| SKU | GTIN |
|---|---|
| L485195-1EA | |
| 685828-5G | 4061832759203 |
| 685828-25G | 4061832759197 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service