632023
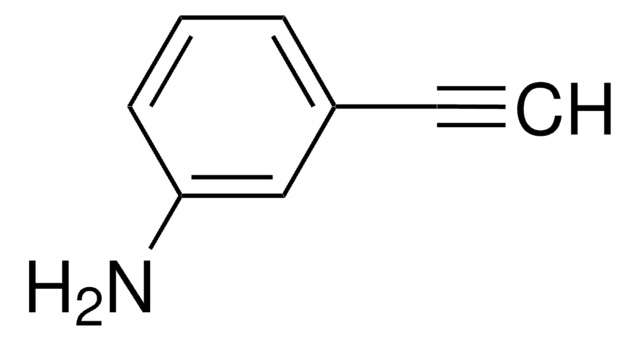
3-Hydroxyphenylacetylene
≥95.0%
Synonym(s):
3-Ethynylphenol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C8H6O
CAS Number:
Molecular Weight:
118.13
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥95.0%
refractive index
n20/D 1.5840 (lit.)
density
1.083 g/mL at 25 °C (lit.)
SMILES string
Oc1cccc(c1)C#C
InChI
1S/C8H6O/c1-2-7-4-3-5-8(9)6-7/h1,3-6,9H
InChI key
AODMJIOEGCBUQL-UHFFFAOYSA-N
Application
3-Hydroxyphenyl acetylene exhibits chromogenic and fluorogenic properties and hence it can be used as a probe for labeling of bacterial enzymes, which degrade toluene.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
217.4 °F
Flash Point(C)
103 °C
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
"Hydration of aromatic terminal alkynes catalyzed by iron (III) sulfate hydrate under chlorine-free conditions"
Bassetti M, et al.
Tetrahedron Letters, 55(9), 1608-1612 (2014)
Mary E Kauffman et al.
Journal of microbiological methods, 55(3), 801-805 (2003-11-11)
3-hydroxyphenylacetylene (3-HPA) served as a novel, activity-dependent, fluorogenic and chromogenic probe for bacterial enzymes known to degrade toluene via meta ring fission of the intermediate, 3-methylcatechol. By this direct physiological analysis, cells grown with an aromatic substrate to induce the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service