480134
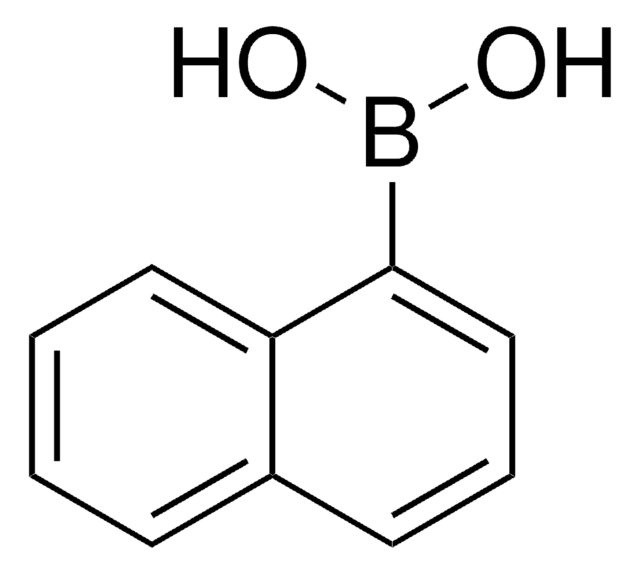
2-Naphthylboronic acid
≥95.0%
Synonym(s):
2-Naphthaleneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
C10H7B(OH)2
CAS Number:
Molecular Weight:
171.99
Beilstein:
2936449
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥95.0%
mp
269-275 °C (lit.)
SMILES string
OB(O)c1ccc2ccccc2c1
InChI
1S/C10H9BO2/c12-11(13)10-6-5-8-3-1-2-4-9(8)7-10/h1-7,12-13H
InChI key
KPTRDYONBVUWPD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Used in a study of an enantioselective rhodium-catalyzed addition of aryl boronic acids to 2,2,2-trifluoroacetophenones leading to chiral, tertiary trifluoromethyl alcohols. Also employed in a study of a palladium-catalyzed addition of aryl boronic acids to nitriles providing aryl ketones and to aryloxy nitriles providing benzofurans.
Other Notes
Contains varying amounts of anhydride
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sébastien L X Martina et al.
Chemical communications (Cambridge, England), (39), 4093-4095 (2006-10-07)
The catalytic asymmetric 1,2-addition of a series of arylboronic acids to 2,2,2-trifluoroacetophenones is described with high isolated yields (up to 96%) and good enantioselectivities (up to 83% ee) using a rhodium(I)/phosphoramidite catalyst.
Jae-Ryung Cha et al.
Journal of fluorescence, 24(4), 1215-1224 (2014-05-27)
Blue light-emitting spiro[benzotetraphene-fluorene] (SBTF)-based host materials, 3-(1-naphthyl)-10-naphthylspiro[benzo[ij]tetraphene-7,9'-fluorene] (1), 3-(2-naphthyl)-10-naphthylspiro[benzo[ij]tetraphene-7,9'-fluorene] (2), and 3-[2-(6-phenyl)naphthyl]-10-naphthylspiro[benzo[ij]tetraphene-7,9'-fluorene] (3) were designed and prepared via multi-step Suzuki coupling reactions. Introducing various aromatic groups into SBTF core lead to a reduction in band gap and a determination of
Saikat Das et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 18(1), 79-86 (2016-10-28)
Nuclear magnetic resonance is applied to investigate the relative positions and interactions between cationic and non-ionic carbohydrate-based surfactants in mixed micelles with D
Baowei Zhao et al.
Organic letters, 8(26), 5987-5990 (2006-12-15)
[Structure: see text] A cationic palladium complex catalyzed addition of arylboronic acids to nitriles to yield aryl ketones with moderate to good yields was developed. A one-step synthesis of benzofurans from phenoxyacetonitriles under the catalysis of [(bpy)Pd+(micro-OH)]2(-OTf)2 or [(bpy)Pd2+(H2O)2](-OTf)2 was
Alessio Innocenti et al.
Bioorganic & medicinal chemistry letters, 19(10), 2642-2645 (2009-04-21)
Inhibition of the beta-carbonic anhydrases (CAs, EC 4.2.1.1) from the pathogenic fungi Cryptococcus neoformans (Can2) and Candida albicans (Nce103) with a series of aromatic, arylalkenyl- and arylalkylboronic acids was investigated. Aromatic, 4-phenylsubstituted- and 2-naphthylboronic acids were the best Can2 inhibitors
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service