411760
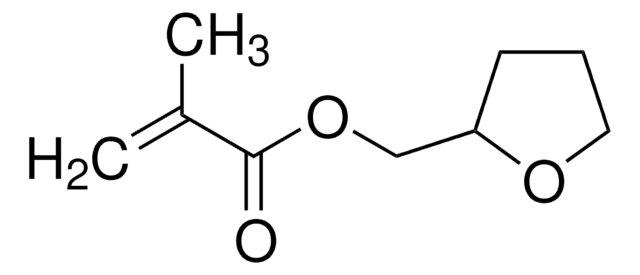
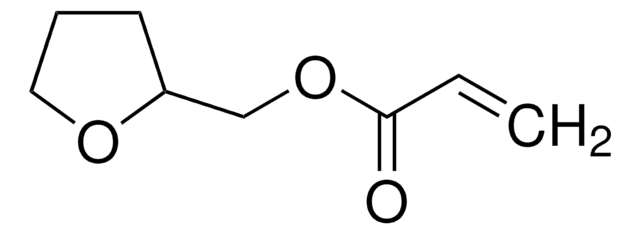
Furfuryl methacrylate
97%, contains 200 ppm monomethyl ether hydroquinone as inhibitor
Synonym(s):
Methacrylic Acid Furfuryl Ester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C9H10O3
CAS Number:
Molecular Weight:
166.17
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Assay
97%
form
liquid
contains
200 ppm monomethyl ether hydroquinone as inhibitor
refractive index
n20/D 1.482 (lit.)
bp
80-82 °C/5 mmHg (lit.)
density
1.078 g/mL at 25 °C (lit.)
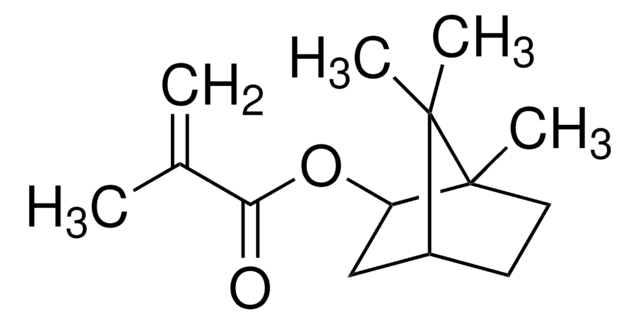
SMILES string
CC(=C)C(=O)OCc1ccco1
InChI
1S/C9H10O3/c1-7(2)9(10)12-6-8-4-3-5-11-8/h3-5H,1,6H2,2H3
InChI key
DWXAVNJYFLGAEF-UHFFFAOYSA-N
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
195.8 °F - closed cup
Flash Point(C)
91 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Sunanda Sain et al.
Polymers, 12(11) (2020-10-30)
The fabrication of smart biocomposites from sustainable resources that could replace today's petroleum-derived polymer materials is a growing field of research. Here, we report preparation of novel biocomposites using nanocellulose networks extracted from food residue (onion skin) and a vegetable
D Zaldívar et al.
Biomaterials, 14(14), 1073-1079 (1993-11-01)
Biocompatible copolymers of N-vinylpyrrolidone (P) and furfuryl methacrylate (F) were prepared by free radical polymerization in N,N-dimethylformamide solution at 50 degrees C, using 2,2'-azobisisobutyronitrile as initiator, at low and high conversion. The microstructure of copolymers prepared at low conversion was
Sunanda Sain et al.
Polymers, 12(2) (2020-01-26)
This work focuses on the development of cross-linked polymer from a highly unsaturated vegetable oil, tung oil (TO) and a bio-based acrylate, furfuryl methacrylate (FMA). The presence of a high degree of unsaturated carbon-carbon bonding in TO makes it a
A Amalin Kavitha et al.
ACS applied materials & interfaces, 1(7), 1427-1436 (2010-04-02)
This investigation reports the effective use of the Diels-Alder (DA) reaction, a "click reaction" in the preparation of thermally amendable and self-healing polymeric materials having reactive furfuryl functionality. In this case, the DA and retro-DA (rDA) reactions were carried out
Sovan Lal Banerjee et al.
Soft matter, 13(47), 9024-9035 (2017-11-28)
Amphiphilic diblock copolymers of poly(furfuryl methacrylate) (PFMA) with cationic poly(2-(methacryloyloxy)ethyltrimethyl ammonium chloride) (PFMA-b-PMTAC) and anionic poly(sodium 4-vinylbenzenesulfonate) (PFMA-b-PSS) were prepared via reversible addition fragmentation chain-transfer (RAFT) polymerization by using PFMA as a macro-RAFT agent. The formation of the block copolymer
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service