All Photos(1)
About This Item
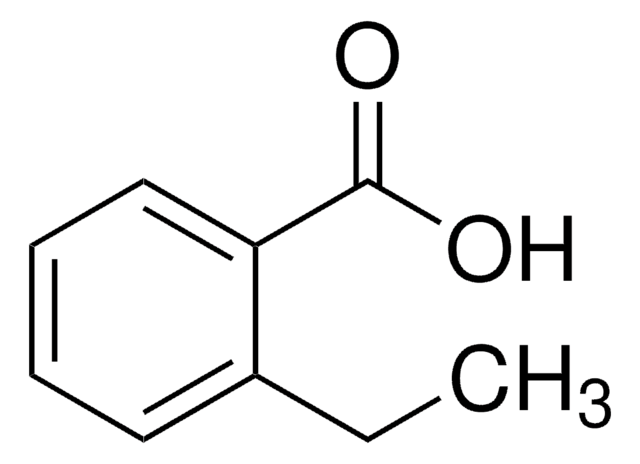
Linear Formula:
C2H5C6H4CO2H
CAS Number:
Molecular Weight:
150.17
Beilstein:
2041840
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
mp
112-113 °C (lit.)
functional group
carboxylic acid
SMILES string
CCc1ccc(cc1)C(O)=O
InChI
1S/C9H10O2/c1-2-7-3-5-8(6-4-7)9(10)11/h3-6H,2H2,1H3,(H,10,11)
InChI key
ZQVKTHRQIXSMGY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
4-Ethylbenzoic acid reacts with lanthanum nitrate in aqueous solution to yield the polymer catena-poly[[aqua(4-ethylbenzoic acid-κO)lanthanum(III)]-tri-μ-4-ethylbenzoato].
Application
4-Ethylbenzoic acid was used in the synthesis of ethyl 4-vinyl-α-cyano-β-phenylcinnamate. It was also used to functionalize the edge of “pristine” graphite in the presence of polyphosphoric acid/phosphorus pentoxide.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Seo-Yoon Bae et al.
ACS nano, 5(6), 4974-4980 (2011-05-20)
We report edge-selective functionalization of graphite (EFG) for the production of large-area uniform graphene films by simply solution-casting EFG dispersions in dichloromethane on silicon oxide substrates, followed by annealing. The resultant graphene films show ambipolar transport properties with sheet resistances
Juan Yang et al.
Acta crystallographica. Section E, Structure reports online, 66(Pt 2), m183-m184 (2010-01-01)
The reaction of lanthanum nitrate and 4-ethyl-benzoic acid (EBAH) in aqueous solution yielded the title polymer, [La(C(9)H(9)O(2))(3)(C(9)H(10)O(2))(H(2)O)](n). The asymmetric unit contains one La(III) atom, three 4-ethyl-benzoate (EBA) ligands, one neutral EBAH ligand and one coordinated water mol-ecule. Each La(III) ion
Functional Polymers. VII. Ethyl 4-Vinyl-α-cyano-β-phenylcinnamate.
Sumida Y and Vogl O.
Polymer Journal, 13(6), 521-536 (1981)
Ruthenium(II)-catalyzed synthesis of hydroxylated arenes with ester as an effective directing group.
Yiqing Yang et al.
Organic letters, 14(11), 2874-2877 (2012-05-16)
An unprecedented Ru(II) catalyzed ortho-hydroxylation has been developed for the facile synthesis of a variety of multifunctionalized arenes from easily accessible ethyl benzoates with ester as an efficient directing group. Both the TFA/TFAA cosolvent system and oxidants serve as the
J L Ramos et al.
Science (New York, N.Y.), 235(4788), 593-596 (1987-01-30)
Increasing quantities of man-made organic chemicals are released each year into the biosphere. Some of these compounds are both toxic and relatively resistant to physical, chemical, or biological degradation, and they thus constitute an environmental burden of considerable magnitude. Genetic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service