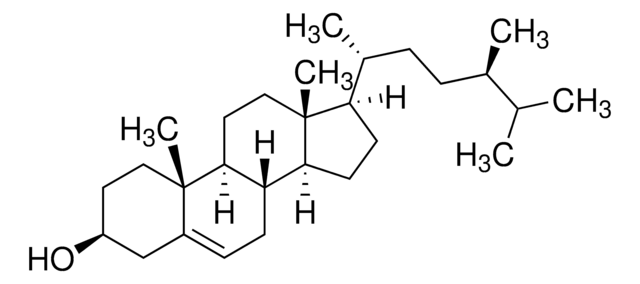
PHL89514
Campesterol
phyproof® Reference Substance
Synonym(s):
24α-Methyl-5-cholesten-3β-ol, 24(R)-Ergost-5-en-3β-ol
About This Item
Recommended Products
biological source
Glycine max (soybean)
grade
primary reference standard
product line
phyproof® Reference Substance
Assay
≥90.0% (GC)
form
crystals
manufacturer/tradename
PhytoLab
storage temp.
2-8°C
SMILES string
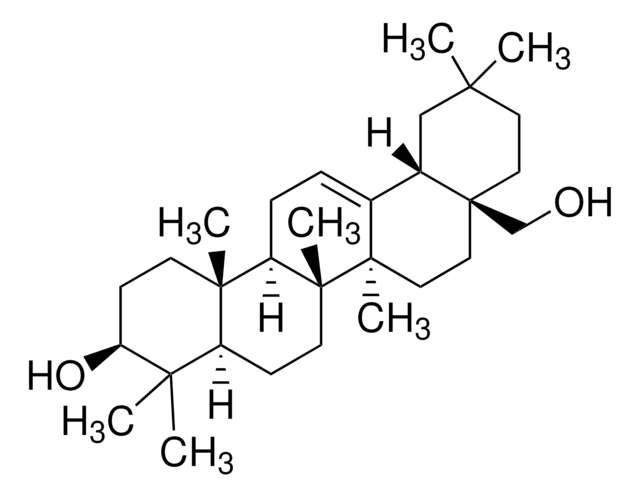
[H][C@@]12CC=C3C[C@@H](O)CC[C@]3(C)[C@@]1([H])CC[C@]4(C)[C@H](CC[C@@]24[H])[C@H](C)CC[C@H](C)C(C)C
InChI
1S/C28H48O/c1-18(2)19(3)7-8-20(4)24-11-12-25-23-10-9-21-17-22(29)13-15-27(21,5)26(23)14-16-28(24,25)6/h9,18-20,22-26,29H,7-8,10-17H2,1-6H3/t19-,20+,22-,23-,24+,25-,26-,27-,28+/m0/s1
InChI key
SGNBVLSWZMBQTH-ZRUUVFCLSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Campesterol is a major phytosterol having a core skeleton similar to cholesterol but with a methyl side chain at C24. It is used in the management of hypercholesterolemia, besides having antioxidant, anti-inflammatory, and anti-cancer properties.
Application
- Development of two high-performance liquid chromatography (HPLC)-diode array ultraviolet/visible detection (DAD-UV/VIS) and liquid chromatography-mass spectrometry (LC-MS) methods to determine campesterol, stigmasterol, and daucosterol from the herbal samples of Artemisia apiacea
- Multi-residue determination of β-sitosterol, campesterol, and stigmasterol in rat plasma samples by liquid chromatography-atmospheric pressure chemical ionization-tandem mass spectrometry (LC-APCI-MS/MS) method
- Ultrasound-assisted extraction (UAE) of campesterol, β-sitosterol, and stigmasterol from passion fruit seed oil samples for their determination by gas chromatography-flame ionization detection (GC-FID)
- Analysis of 119 vegetable oil samples from 7 different varieties to detect and determine squalene and four sterols using a developed and validated non-destructive method based on proton nuclear magnetic resonance (1H NMR) spectroscopy combined with partial least squares (PLS) method
- Multi-residue analysis of isoquercitrin, campesterol, emodin 8-O-β-D-glucopyranoside, and quercetin in dried stem and flower extracts of Reynoutria sachalinensis by high-performance liquid chromatography coupled with diode-array detection
Other Notes
Legal Information
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service