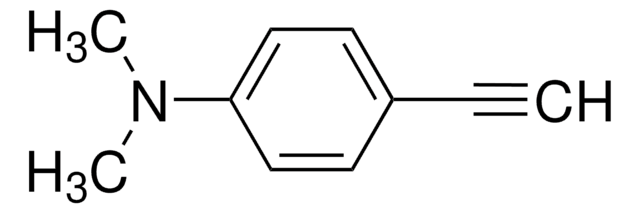
404330
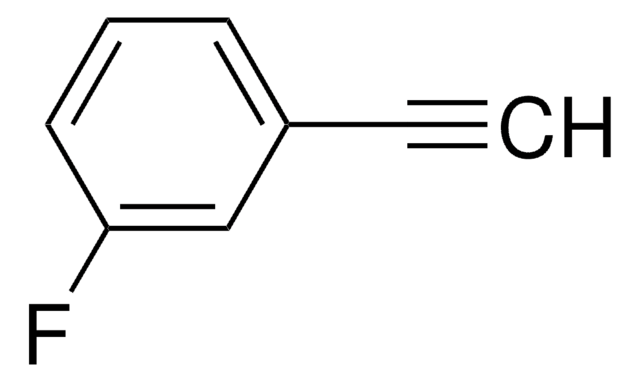
1-Ethynyl-4-fluorobenzene
99%
Synonym(s):
(4-Fluorophenyl)acetylene, (4-Fluorophenyl)ethyne, (p-Fluorophenyl)acetylene, (p-Fluorophenyl)ethyne, 1-Fluoro-4-ethynylbenzene, 2-(4-Fluorophenyl)acetylene, 4-Ethynylfluorobenzene, 4-Fluoroethynylbenzene
About This Item
Recommended Products
Quality Level
Assay
99%
form
solid
refractive index
n20/D 1.516 (lit.)
bp
55-56 °C/40 mmHg (lit.)
mp
26-27 °C (lit.)
density
1.048 g/mL at 25 °C (lit.)
functional group
fluoro
storage temp.
2-8°C
SMILES string
Fc1ccc(cc1)C#C
InChI
1S/C8H5F/c1-2-7-3-5-8(9)6-4-7/h1,3-6H
InChI key
QXSWHQGIEKUBAS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- 3-(4-Fluorophenyl)-1H-isochromen-1-one by reacting with 2-iodobenzoic acid in the presence of copper catalyst.
- 4-Bromo-3,5-difluoro-2,6-bis-(4-fluoro-phenylethynyl)-pyridine by reacting with 2,4,6-tribromo-3,5-difluoropyridine by palladium catalyzed Sonogashira reaction.
- 1-(4-Fluorophenyl)-3-methoxy-3-(4-methoxyphenyl)propan-1-one by reacting with p-anisaldehyde.
- 5-(4-Fluorophenyl)-1-(2-nitrobenzyl)-1H-1,2,3-triazole by reacting with 1-(bromomethyl)-2-nitrobenzene in the presence of sodium azide.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Sol. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
4.1B - Flammable solid hazardous materials
WGK
WGK 3
Flash Point(F)
84.2 °F - closed cup
Flash Point(C)
29 °C - closed cup
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 404330-5G | 4061831986822 |
| 404330-100MG | |
| 404330-1G | 4061831986815 |
| 404330-500MG |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service