122831
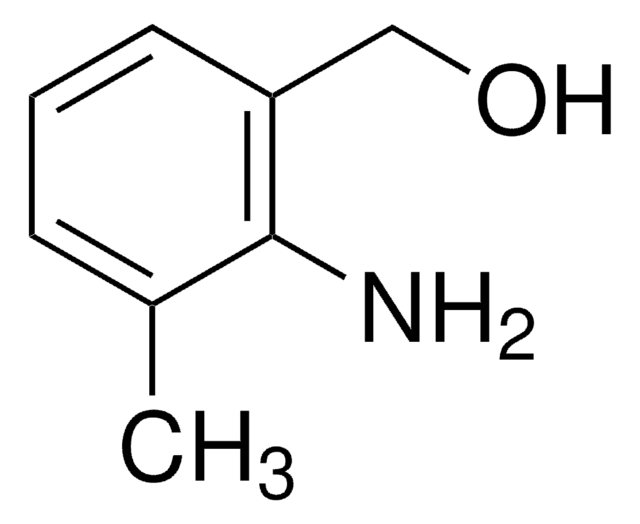
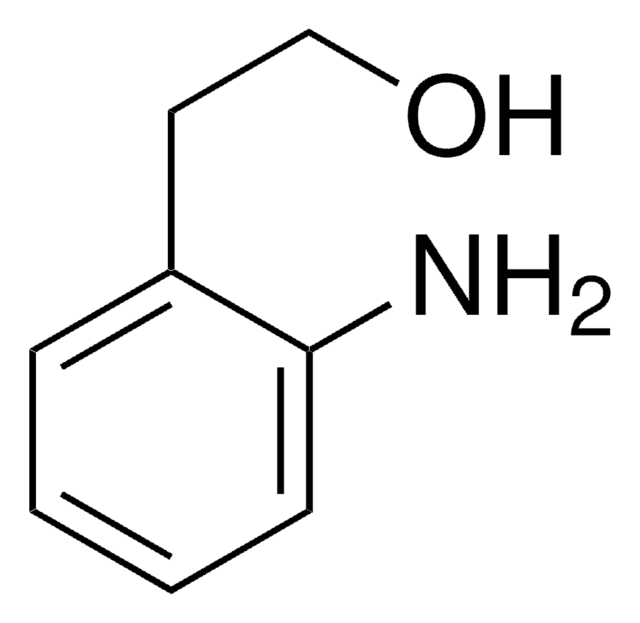
2-Aminobenzyl alcohol
98%
Synonym(s):
2-(Hydroxymethyl)aniline
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
H2NC6H4CH2OH
CAS Number:
Molecular Weight:
123.15
Beilstein:
1072211
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
bp
162 °C/15 mmHg (lit.)
mp
81-83 °C (lit.)
functional group
hydroxyl
SMILES string
Nc1ccccc1CO
InChI
1S/C7H9NO/c8-7-4-2-1-3-6(7)5-9/h1-4,9H,5,8H2
InChI key
VYFOAVADNIHPTR-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Aminobenzyl alcohol is oxidatively cyclised with an array of ketones in dioxane at 80°C in the presence of a ruthenium catalyst and KOH to give corresponding quinolines. It undergoes oxidation catalyzed by heterotrimetallic RuMnMn species on the hydrotalcite surface in the presence of O2 to yield 2-aminobenzaldehyde.
Application
2-Aminobenzyl alcohol was used in the synthesis of ethyl 2-hydroxymethylcarbanilate.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Heterotrimetallic RuMnMn species on a hydrotalcite surface as highly efficient heterogeneous catalysts for liquid-phase oxidation of alcohols with molecular oxygen.
Kohki Ebitani et al.
Angewandte Chemie (International ed. in English), 44(22), 3423-3426 (2005-04-30)
Facile intramolecular nucleophilic attack by alkoxide ions on ethyl and p-nitrophenyl carbamates.
J E Hutchins et al.
Journal of the American Chemical Society, 95(11), 3786-3790 (1973-05-30)
Ruthenium-catalysed oxidative cyclisation of 2-aminobenzyl alcohol with ketones: modified Friedlaender quinoline synthesis.
Cho CS, et al.
Chemical Communications (Cambridge, England), 24, 2576-2577 (2001)
N Sundaraganesan et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 61(3), 377-385 (2004-12-08)
The Fourier transform Raman and Fourier transform infrared spectra of 2-aminobenzyl alcohol (2ABA) were recorded in the solid phase. Geometry optimizations were done with out any constraint and harmonic vibrational wave numbers and several thermodynamic parameters were calculated for the
J P Chism et al.
Chemical research in toxicology, 2(3), 150-156 (1989-05-01)
Previous results have suggested that key intermediates in the activation of 2-nitrotoluene and 2,6-dinitrotoluene are 2-aminobenzyl alcohol and 2-amino-6-nitrobenzyl alcohol, respectively. In order to determine the metabolic pathway(s) involved in the activation steps, calf thymus DNA and [14C]-2-aminobenzyl alcohol or
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service