T54801
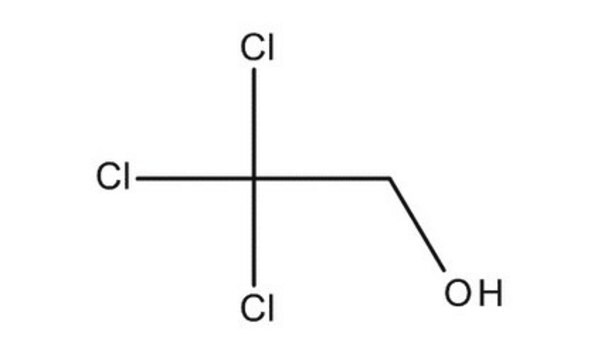
2,2,2-Trichloroethanol
ReagentPlus®, ≥99%
Synonym(s):
Trichloroethyl alcohol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
Cl3CCH2OH
CAS Number:
Molecular Weight:
149.40
Beilstein:
1697495
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
vapor density
5.16 (vs air)
vapor pressure
1 mmHg ( 20 °C)
product line
ReagentPlus®
Assay
≥99%
refractive index
n20/D 1.49 (lit.)
bp
152-154 °C (lit.)
mp
17.8 °C (lit.)
density
1.56 g/mL at 20 °C (lit.)
SMILES string
OCC(Cl)(Cl)Cl
InChI
1S/C2H3Cl3O/c3-2(4,5)1-6/h6H,1H2
InChI key
KPWDGTGXUYRARH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
The 2,2,2-Trichloroethyl (TCE) moiety of 2,2,2-trichloroethanol is mainly used as a protecting group for acids, acetals and also phosphoryl groups of nucleotides. It can be easily cleaved by zinc reduction.
Some of its other application include:
Some of its other application include:
- Conversion of per-O-acetylated sugars to per-O-acetylated trichloroethyl derivatives.
- Preparation of TCE ester of chlorosulfuric acid.
- Preparation of 2,2,2-trichloroethyl acetimidate hydrochloride.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - STOT SE 3
Target Organs
Central nervous system
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
`BF3-etherate induced formation of 2, 2, 2-trichloroethyl glycopyranosides. Selective visualization of carbohydrate derivatives on TLC plates.
Magnusson, et al.
Acta Chemica Scandinavica. Series B, 35(3) (1981)
2, 2, 2-Trichloroethyl chloroformate: a general reagent for demethylation of tertiary methylamines.
Montzka TA, et al.
Tetrahedron Letters, 15(14), 1325-1327 (1974)
Synthesis and protection of aryl sulfates using the 2, 2, 2-trichloroethyl moiety.
Liu Y, et al.
Organic Letters, 6(2), 209-212 (2004)
e-EROS Encyclopedia of Reagents for Organic Synthesis null
Preparation of N1?Phenylacetamidine 4?Bromobenzoate Using 2, 2, 2?Trichloroethyl Acetimidate Hydrochloride.
Wei L & Caron S.
Organic Syntheses, 174-181 null
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service