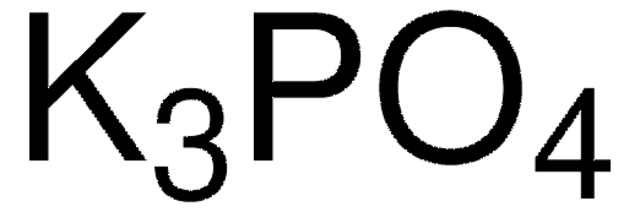597988
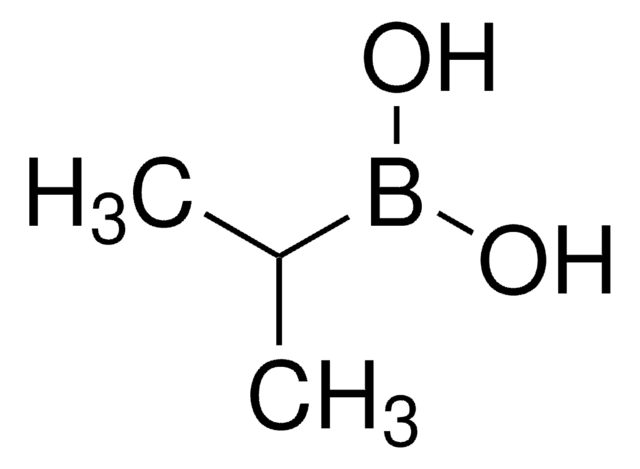
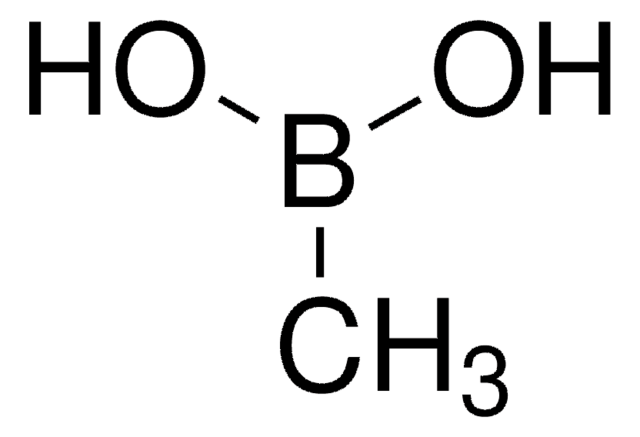
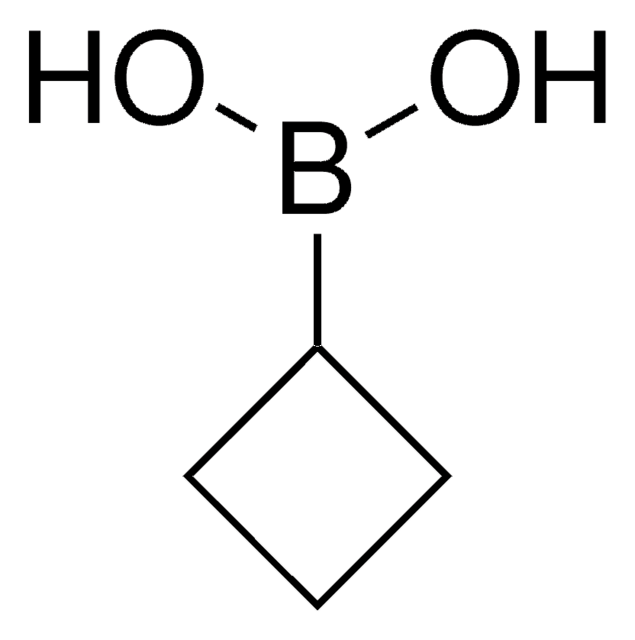
Cyclopropylboronic acid
Synonym(s):
Cyclopropaneboronic acid
About This Item
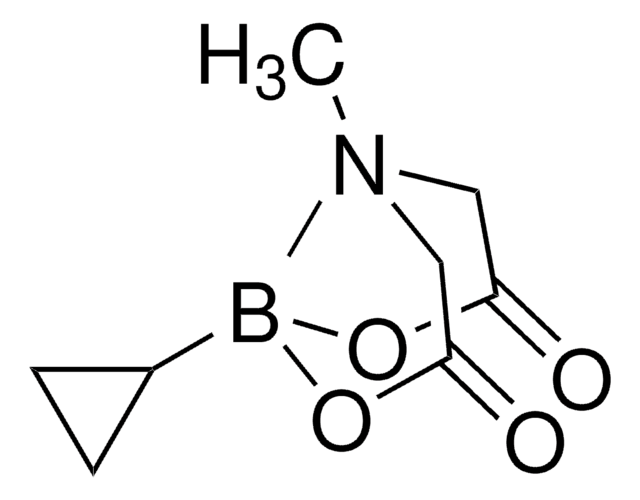
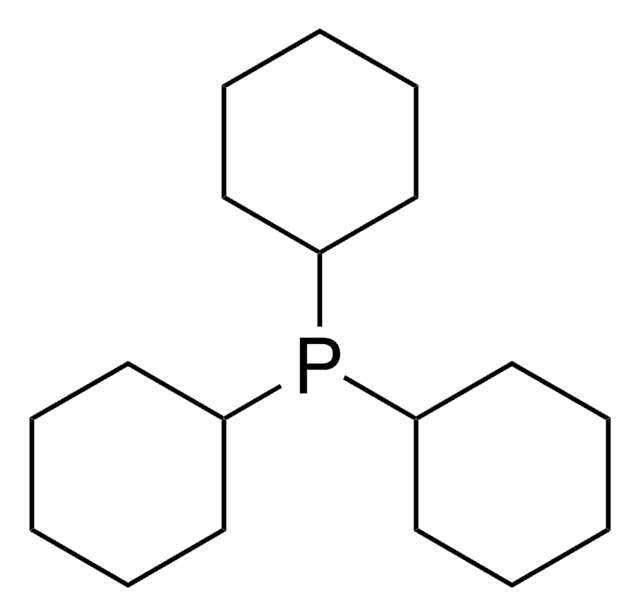
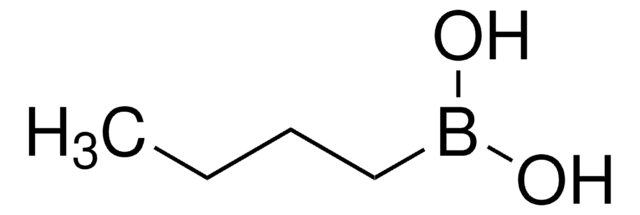
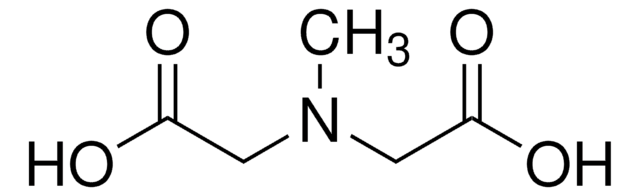
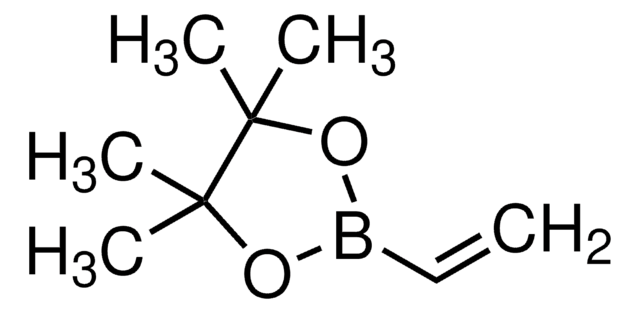
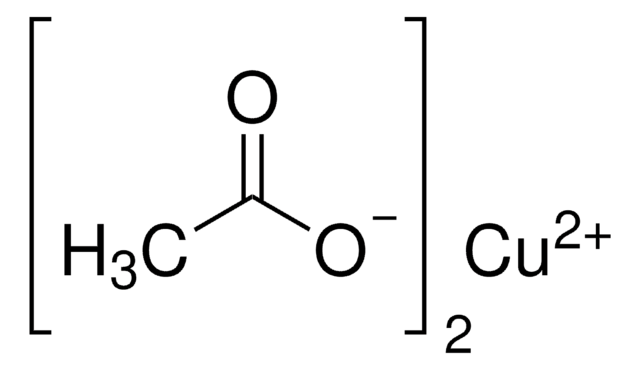
Recommended Products
form
solid
mp
90-95 °C (lit.)
storage temp.
−20°C
SMILES string
OB(O)C1CC1
InChI
1S/C3H7BO2/c5-4(6)3-1-2-3/h3,5-6H,1-2H2
InChI key
WLVKDFJTYKELLQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Microwave-assisted copper(II)-catalyzed N-cyclopropylation
- Nickel- and copper-catalyzed Suzuki-Miyaura coupling reaction of arenes
- Palladacycle-catalyzed Suzuki-cross coupling of aryl halides with cyclopropylboronic acid
- Palladium(0)-catalyzed cyclopropane C-H bond functionalization
- Palladium-catalyzed decarboxylative coupling
- Palladium-catalyzed ligand-directed oxidative functionalization of cyclopropanes
- Palladium-catalyzed Suzuki coupling reaction
Reagent used in Preparation of
- Diaryl ketones by arylation of arylboronic acids with aromatic aldehydes catalyzed by Cu(OTf)2 and Xantphos
- Aminothiazolylpyrrolidine-based tartrate diamides as TACE inhibitors to treat inflammatory disorders and cancer
Other Notes
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Repr. 1B
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)