565415
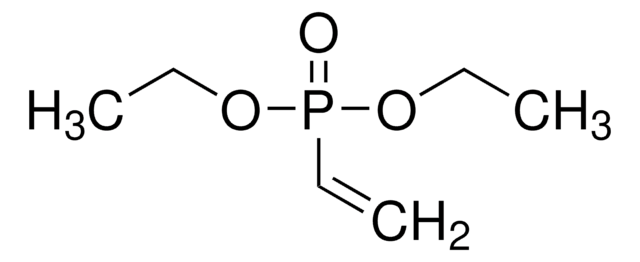
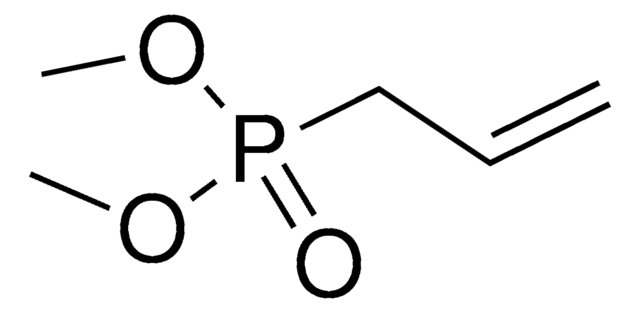
Diethyl allylphosphonate
98%
Synonym(s):
2-Propenyl-phosphonic acid diethyl ester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH2=CHCH2P(O)(OC2H5)2
CAS Number:
Molecular Weight:
178.17
MDL number:
UNSPSC Code:
12352108
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
reaction suitability
reaction type: C-C Bond Formation
refractive index
n20/D 1.4340 (lit.)
bp
46 °C/0.35 mmHg (lit.)
bulk density
1.022 g/mL
functional group
phosphonate
SMILES string
CCOP(=O)(CC=C)OCC
InChI
1S/C7H15O3P/c1-4-7-11(8,9-5-2)10-6-3/h4H,1,5-7H2,2-3H3
InChI key
YPJHXRAHMUKXAE-UHFFFAOYSA-N
Related Categories
Application
Reactant for:
- Copolymerization of phosphonated allyl monomers and maleic anhydride
- Enantioselective synthesis of solamin type mono-THF acetogenins
- RCM reaction yielding oxaphospholene and oxaphosphinene heterocycles
- Synthesis of spongistatin 2 using Wittig coupling
- Stereoselective synthesis of pentacyclic furanosteroids
- Preparation of protected polyhydroxylated β -amino acid constitutents of microsclerodermins
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Elizabeth I Parkinson et al.
Nature chemical biology, 15(11), 1049-1056 (2019-08-28)
Fosmidomycin and related molecules comprise a family of phosphonate natural products with potent antibacterial, antimalarial and herbicidal activities. To understand the biosynthesis of these compounds, we characterized the fosmidomycin producer, Streptomyces lavendulae, using biochemical and genetic approaches. We were unable
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service