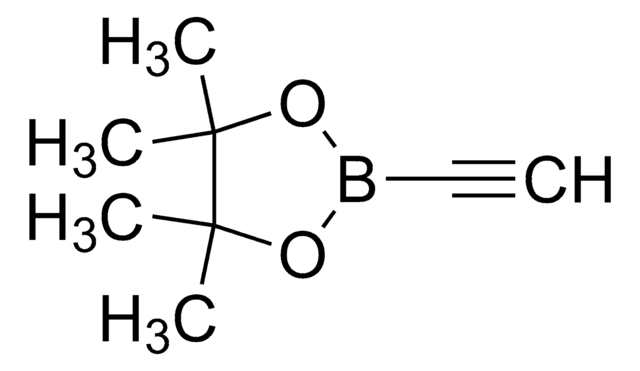
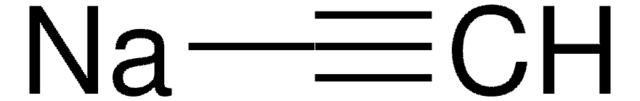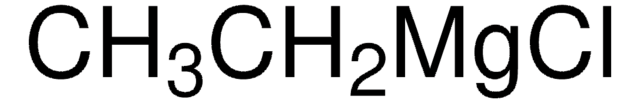346152
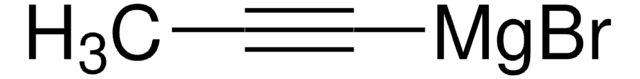
Ethynylmagnesium bromide solution
0.5 M in THF
Synonym(s):
Acetylenemagnesium bromide, Bromoethynylmagnesium, Ethynylmagnesium bromide
About This Item
Recommended Products
Quality Level
reaction suitability
reaction type: Grignard Reaction
concentration
0.5 M in THF
density
0.94 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
Br[Mg]C#C
InChI
1S/C2H.BrH.Mg/c1-2;;/h1H;1H;/q;;+1/p-1
InChI key
HUGJUYPSXULVQQ-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Grafting of aliphatic polymer.
- Synthesis of anthracene-containing species like 1,8-dichloro-10-(ethynyl)anthracene.
- The preparation of the alkyne component in an enyne cross-metathesis leading to conjugated dienes.
- Ethynylation of chiral α-(dibenzylamino) aldehydes.
- The synthesis of ethynyl aziridines.
- To prepare alkyl(triethynyl)germanes from tert-alkyltrichlorogermanes and 2-butyltrichlorogermane.
- The preperation of a key intermediate in the total synthesis of (+)-4-demethoxydaunomycin.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Carc. 2 - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B - STOT SE 3 - Water-react 2
Target Organs
Respiratory system
Supplementary Hazards
Storage Class Code
4.3 - Hazardous materials which set free flammable gases upon contact with water
WGK
WGK 3
Flash Point(F)
-20.2 °F
Flash Point(C)
-29 °C
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service