All Photos(1)
About This Item
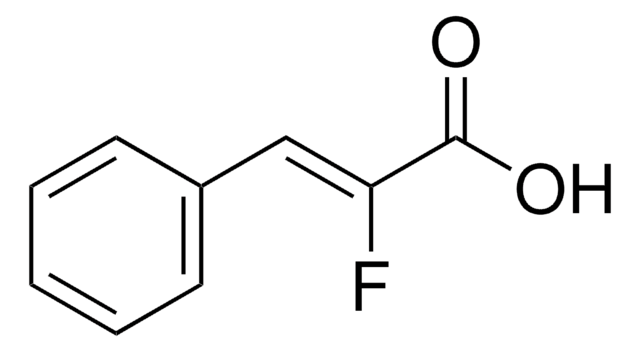
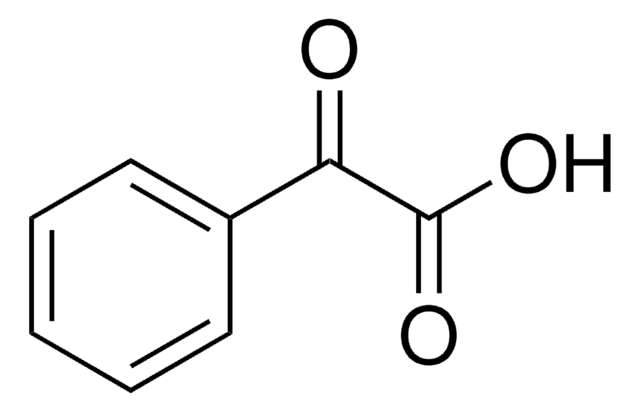
Linear Formula:
FC6H4CH=CHCO2H
CAS Number:
Molecular Weight:
166.15
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
mp
178-180 °C (lit.)
SMILES string
OC(=O)\C=C\c1ccccc1F
InChI
1S/C9H7FO2/c10-8-4-2-1-3-7(8)5-6-9(11)12/h1-6H,(H,11,12)/b6-5+
InChI key
IOUDZAFBPDDAMK-AATRIKPKSA-N
Application
2-Fluorocinnamic acid was used in the synthesis of acrylamide.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Alexandre L'Heureux et al.
Bioorganic & medicinal chemistry letters, 15(2), 363-366 (2004-12-18)
Replacement of the morpholinyl moiety in (S,E)-N-[1-(3-morpholinophenyl)ethyl]-3-phenylacrylamide (1) with heteroaryl groups led to the identification of (S,E)-N-1-[3-(6-fluoropyridin-3-yl)phenyl]ethyl-3-(2-fluorophenyl)acrylamide (5) as a potent KCNQ2 potassium channel opener. Among this series of heteroaryl substituted acrylamides, (S,E)-N-1-[3-(1H-pyrazol-1-yl)phenyl]ethyl-3-(2-fluorophenyl)acrylamide (9) exhibits balanced potency and efficacy. The
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service