A44207
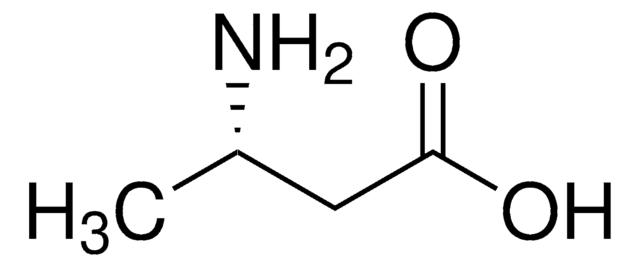
3-Aminobutanoic acid
97%, for peptide synthesis
Synonym(s):
(±)-3-Aminobutyric acid, DL-3-Aminobutyric acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3CH(NH2)CH2COOH
CAS Number:
Molecular Weight:
103.12
Beilstein:
1720563
EC Number:
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Product Name
3-Aminobutanoic acid, 97%
Quality Level
Assay
97%
form
powder and chunks
reaction suitability
reaction type: solution phase peptide synthesis
color
white
mp
189 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
CC(N)CC(O)=O
InChI
1S/C4H9NO2/c1-3(5)2-4(6)7/h3H,2,5H2,1H3,(H,6,7)
InChI key
OQEBBZSWEGYTPG-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
3-Aminobutanoic acid can be used as a reactant to synthesize:
- N
- -aryl amino butanoic acids by Ullmann type aryl amination reaction with aryl halides.
- 3-amino butanoic acid methyl ester, which is used as a chemical intermediate to prepare substituted piperidinone via an ester-imine derivative of aminobutanoic acid.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
CuI-catalyzed coupling reaction of β-amino acids or esters with aryl halides at temperature lower than that employed in the normal Ullmann reaction. Facile synthesis of SB-214857
Ma Dawei and Xia Chengfeng
Organic Letters, 3(16), 2583-2586 (2001)
Roland E Schwarzenbacher et al.
Molecular plant, 13(10), 1455-1469 (2020-07-28)
External and internal signals can prime the plant immune system for a faster and/or stronger response to pathogen attack. β-aminobutyric acid (BABA) is an endogenous stress metabolite that induces broad-spectrum disease resistance in plants. BABA perception in Arabidopsis is mediated
William Kay et al.
The FEBS journal, 273(13), 3002-3013 (2006-06-09)
Lipopolysaccharide (LPS) and beta-glucan from Francisella victoria, a fish pathogen and close relative of highly virulent mammal pathogen Francisella tularensis, have been analyzed using chemical and spectroscopy methods. The polysaccharide part of the LPS was found to contain a nonrepetitive
S Hodge et al.
Bulletin of entomological research, 95(5), 449-455 (2005-10-04)
DL-beta-aminobutyric acid (BABA) is a non-protein amino acid that is an effective inducer of resistance against a variety of plant pathogens. However, examples of BABA-induced resistance against insect herbivores have not been reported. We applied BABA as a soil drench
Mollah Md Hamiduzzaman et al.
Molecular plant-microbe interactions : MPMI, 18(8), 819-829 (2005-09-02)
beta-Aminobutyric acid (BABA) was used to induce resistance in grapevine (Vitis vinifera) against downy mildew (Plasmopara viticola). This led to a strong reduction of mycelial growth and sporulation in the susceptible cv. Chasselas. Comparing different inducers, the best protection was
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service