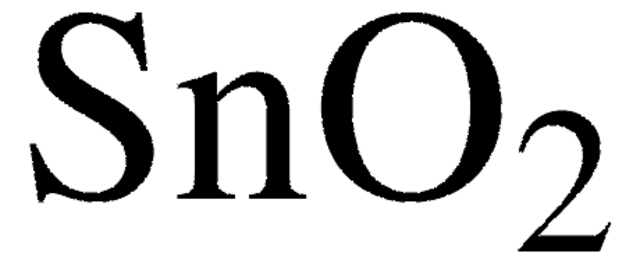549657
Tin(IV) oxide
nanopowder, ≤100 nm avg. part. size
Synonym(s):
Tin oxide, Stannic oxide
About This Item
Recommended Products
form
nanopowder
Quality Level
greener alternative product characteristics
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
avg. part. size
≤100 nm
density
6.95 g/mL at 25 °C (lit.)
application(s)
battery manufacturing
greener alternative category
SMILES string
O=[Sn]=O
InChI
1S/2O.Sn
InChI key
XOLBLPGZBRYERU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Storage Class Code
11 - Combustible Solids
WGK
nwg
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Synthesis, Properties, and Applications of Perovskite-Phase Metal Oxide Nanostructures
Increasing fuel costs and concerns about greenhouse gas emissions have spurred the growth in sales of hybrid electric vehicles (HEVs) that carry a battery pack to supplement the performance of the internal combustion engine (ICE).
Nanomaterials for Energy Storage in Lithium-ion Battery Applications
Recent demand for electric and hybrid vehicles, coupled with a reduction in prices, has caused lithium-ion batteries (LIBs) to become an increasingly popular form of rechargeable battery technology.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service