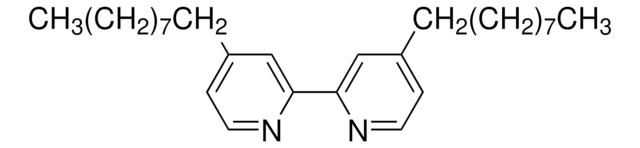
369497
N,N,N′,N′′,N′′-Pentamethyldiethylenetriamine
99%
Synonym(s):
PMDETA, PMDTA
About This Item
Recommended Products
vapor pressure
0.23 mmHg ( 20 °C)
Assay
99%
form
liquid
autoignition temp.
311 °F
expl. lim.
5.6 %
refractive index
n20/D 1.442 (lit.)
bp
198 °C (lit.)
mp
−20 °C (lit.)
density
0.83 g/mL at 25 °C (lit.)
SMILES string
CN(C)CCN(C)CCN(C)C
InChI
1S/C9H23N3/c1-10(2)6-8-12(5)9-7-11(3)4/h6-9H2,1-5H3
InChI key
UKODFQOELJFMII-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- A catalyst in the synthesis of multifunctional silicone acrylate prepolymers for use in UV-curable coatings.
- A multifunctional initiating and cross-linking agent in the synthesis of polyacrylamide hydrogels. It improves the mechanical properties of the hydrogels, such as toughness and resilience, without compromising their biocompatibility.
- An organocatalyst in ring-opening polymerization (ROP) of trimethylene carbonate. This catalyst can be easily removed after the reaction compared to metal catalysts.
- A catalyst along with CuBr to synthesize a series of side-chain azobenzene poly(meth)acrylates via the atom transfer radical polymerization(ATRP) technique.
- An initiator to grow polystyrene chains to prepare polyolefin-polystyrene copolymers.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flash Point(F)
168.8 °F - closed cup
Flash Point(C)
76 °C - closed cup
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Tools and techniques for performing atom transfer radical polymerization (ATRP) with benefits and limitations.
Atom transfer radical polymerization (ATRP) has emerged as one of the most successful synthetic techniques for the preparation of polymers with predetermined molecular weights, narrow molecular weight distributions, and high degrees of chain end functionalities.
ATRP polymerization, chain transfer agent, living polymerization, functional telechelic polymers
Find how atom transfer radical polymerization (ATRP) tools can be used for the synthesis of well-defined functionalized polymers.
Protocols
We presents an article featuring procedures that describe polymerization of methyl methacrylate and vinyl acetate homopolymers and a block copolymer as performed by researchers at CSIRO.
Sigma-Aldrich presents an article about RAFT, or Reversible Addition/Fragmentation Chain Transfer, which is a form of living radical polymerization.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![Tris[2-(dimethylamino)ethyl]amine 97%](/deepweb/assets/sigmaaldrich/product/structures/695/792/ee0ff167-22a3-43a7-83a1-6c4908adf0ae/640/ee0ff167-22a3-43a7-83a1-6c4908adf0ae.png)






![Bis[2-(N,N-dimethylamino)ethyl] ether 97%](/deepweb/assets/sigmaaldrich/product/structures/372/323/505a46ae-b067-4177-8e5f-19a3f4ef9c44/640/505a46ae-b067-4177-8e5f-19a3f4ef9c44.png)