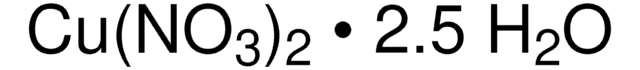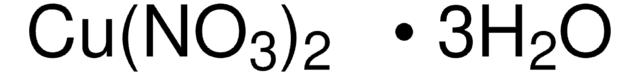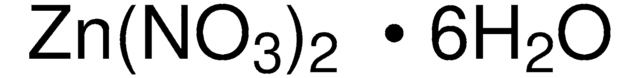203874
Nickel(II) nitrate hexahydrate
99.999% trace metals basis
Synonym(s):
Nickel dinitrate hexahydrate, Nickel nitrate hexahydrate, Nickel(2+) dinitrate hexahydrate, Nickelous nitrate hexahydrate
About This Item
Recommended Products
Assay
99.999% trace metals basis
form
solid
impurities
≤15.0 ppm Trace Metal Analysis
mp
56 °C (lit.)
density
2.05 g/mL at 25 °C (lit.)
application(s)
battery manufacturing
SMILES string
O.O.O.O.O.O.[Ni++].[O-][N+]([O-])=O.[O-][N+]([O-])=O
InChI
1S/2NO3.Ni.6H2O/c2*2-1(3)4;;;;;;;/h;;;6*1H2/q2*-1;+2;;;;;;
InChI key
AOPCKOPZYFFEDA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- As an additive in the fabrication of cellulose acetate polymers(CA) to control the porosity of the polymer. The formation of Ni(NO3)2.6H2O aggregates in polymer matrix during solidification and the strong interaction between Ni, nitrate, and water molecules result in the formation of well-defined pores on the surface of the CA matrix.
- As a starting material to prepare nickel(ii) Schiff base, which is used as a precursor to synthesize NiO nanoparticles by solid-state thermal decomposition method.
- As a dopant to prepare Ni-Ceria catalyst for selective hydrogenation of acetylene.
- As a catalyst to prepare 3,4-dihydropyrimidinone derivatives via Biginelli cyclocondensation.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1A - Eye Dam. 1 - Muta. 2 - Ox. Sol. 2 - Repr. 1B - Resp. Sens. 1 - Skin Irrit. 2 - Skin Sens. 1 - STOT RE 1 Inhalation
Storage Class Code
5.1B - Oxidizing hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Lithium-Ion Battery Performance: Dependence on Material Synthesis and Post‑Treatment Methods
Thermoelectric Performance of Perovskite-type Oxide Materials
The prevailing strategies for heat and electric-power production that rely on fossil and fission fuels are having a negative impact on the environment and on our living conditions.
Plasmonic nanoparticles have unique optical properties that can be tailored to suit a variety of applications in the biotechnology1–8 and electronics9–16 industries.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service