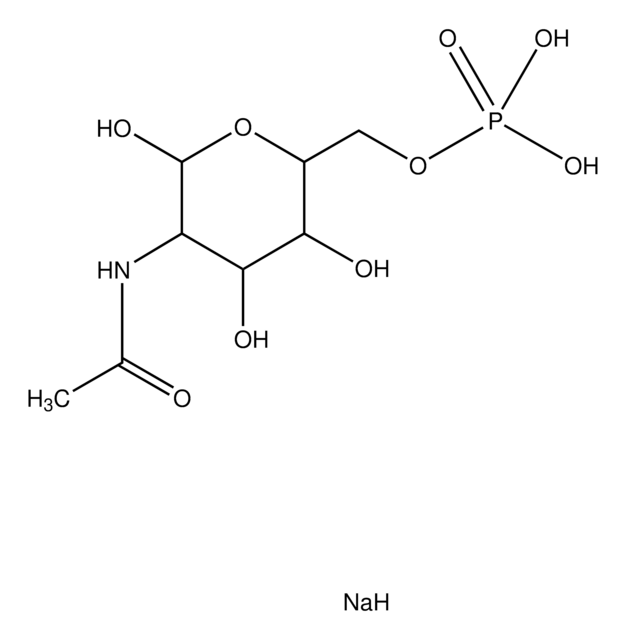
U5252
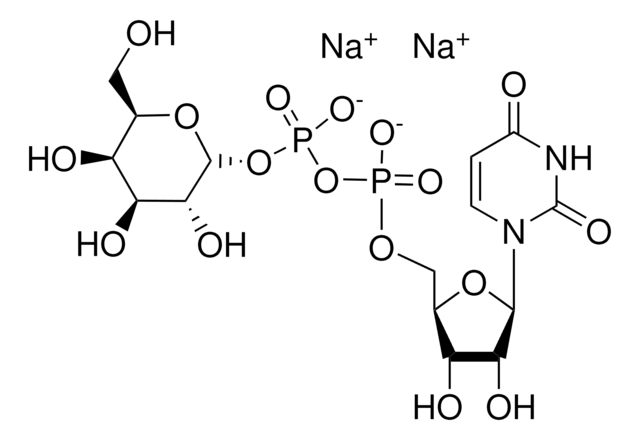
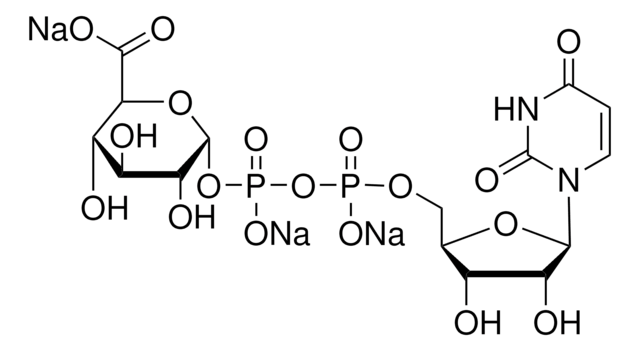
Uridine 5′-diphospho-N-acetylgalactosamine disodium salt
≥97%
Sinónimos:
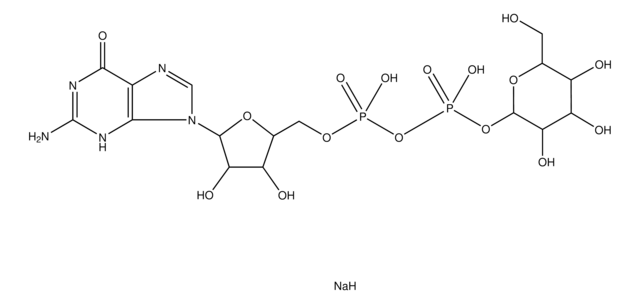
UDP-N-acetylgalactosamine
About This Item
Productos recomendados
biological source
synthetic (organic)
Quality Level
assay
≥97%
form
powder
storage temp.
−20°C
SMILES string
[Na+].[Na+].CC(=O)N[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1OP([O-])(=O)OP([O-])(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)N3C=CC(=O)NC3=O
InChI
1S/C17H27N3O17P2.2Na/c1-6(22)18-10-13(26)11(24)7(4-21)35-16(10)36-39(31,32)37-38(29,30)33-5-8-12(25)14(27)15(34-8)20-3-2-9(23)19-17(20)28;;/h2-3,7-8,10-16,21,24-27H,4-5H2,1H3,(H,18,22)(H,29,30)(H,31,32)(H,19,23,28);;/q;2*+1/p-2/t7-,8-,10-,11+,12-,13-,14-,15?,16?;;/m1../s1
InChI key
HXWKMJZFIJNGES-QCVFHWOISA-L
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
The presence of multiple functional groups and stereocenters in complex carbohydrates makes them challenging targets for the organic chemist.
Glycosyltransferases were initially considered to be specific for a single glycosyl donor and acceptor, which led to the one enzyme-one linkage concept. Subsequent observations have refuted the theory of absolute enzymatic specificity by describing the transfer of analogs of some nucleoside mono- or diphosphate sugar donors.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico