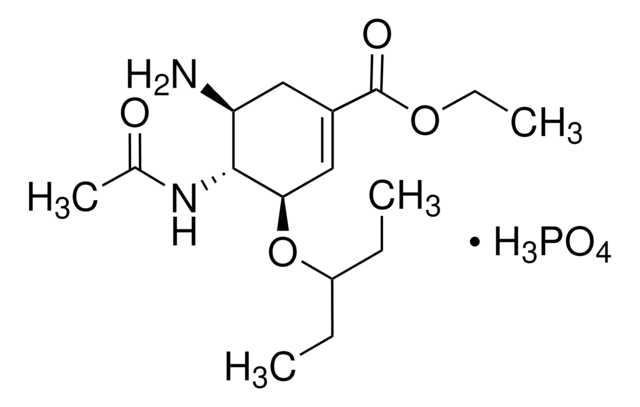
SML0492
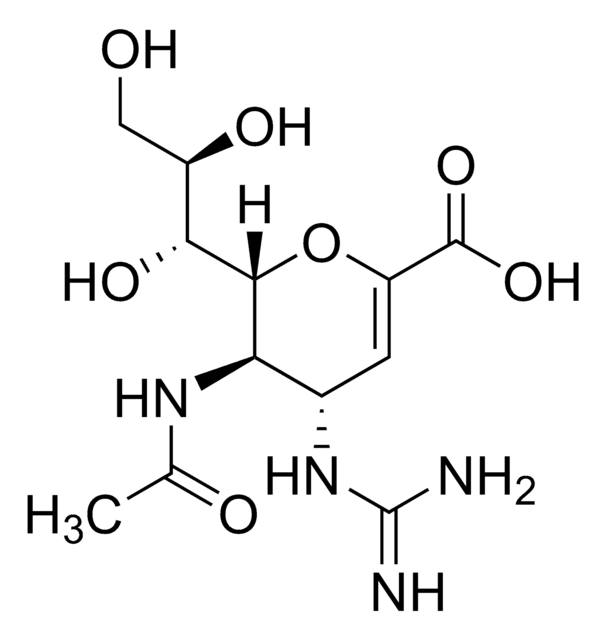
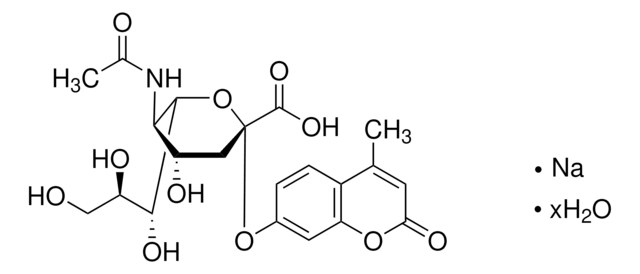
Zanamivir
≥98% (HPLC)
Sinónimos:
4-Guanidino-2,4-dideoxy-2,3-dehydro-N-acetylneuraminic acid, 4-Guanidino-Neu5Ac2en
About This Item
Productos recomendados
assay
≥98% (HPLC)
form
powder
optical activity
[α]/D +30 to +40°, c = 1 in H2O
color
white to beige
solubility
H2O: 10 mg/mL, clear
storage temp.
room temp
SMILES string
CC(=O)N[C@@H]1[C@@H](NC(N)=N)C=C(O[C@H]1[C@H](O)[C@H](O)CO)C(O)=O
InChI
1S/C12H20N4O7/c1-4(18)15-8-5(16-12(13)14)2-7(11(21)22)23-10(8)9(20)6(19)3-17/h2,5-6,8-10,17,19-20H,3H2,1H3,(H,15,18)(H,21,22)(H4,13,14,16)/t5-,6+,8+,9+,10+/m0/s1
InChI key
ARAIBEBZBOPLMB-UFGQHTETSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- as a neuraminidase inhibitor to study its effects on influenza A virus (IAV) like particles
- as a neuraminidase inhibitor to study its effects on lipopolysaccharide (LPS)-mediated pathology in sepsis
- as an antiviral to study its effects on influenza B virus by plaque reduction assay
Biochem/physiol Actions
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico