R0531
Ramatroban
≥98% (HPLC), solid
Sinónimos:
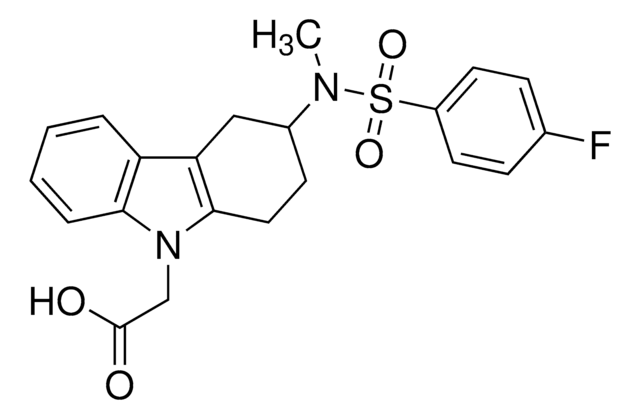
(+)-(3R)-3-[[(4-Fluorophenyl)sulfonyl]amino]-1,2,3,4-tetrahydro-9H-carbazole-9-propanoic acid, BAY u3405
About This Item
Productos recomendados
Quality Level
assay
≥98% (HPLC)
form
solid
color
white
solubility
DMSO: ≥40 mg/mL
H2O: insoluble
originator
Bayer
storage temp.
2-8°C
SMILES string
OC(=O)CCn1c2CC[C@H](Cc2c3ccccc13)NS(=O)(=O)c4ccc(F)cc4
InChI
1S/C21H21FN2O4S/c22-14-5-8-16(9-6-14)29(27,28)23-15-7-10-20-18(13-15)17-3-1-2-4-19(17)24(20)12-11-21(25)26/h1-6,8-9,15,23H,7,10-13H2,(H,25,26)/t15-/m1/s1
InChI key
LDXDSHIEDAPSSA-OAHLLOKOSA-N
Biochem/physiol Actions
Features and Benefits
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Contenido relacionado
Discover Bioactive Small Molecules for Lipid Signaling Research
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico